Posted inDiabetes & Endocrinology news Public Health
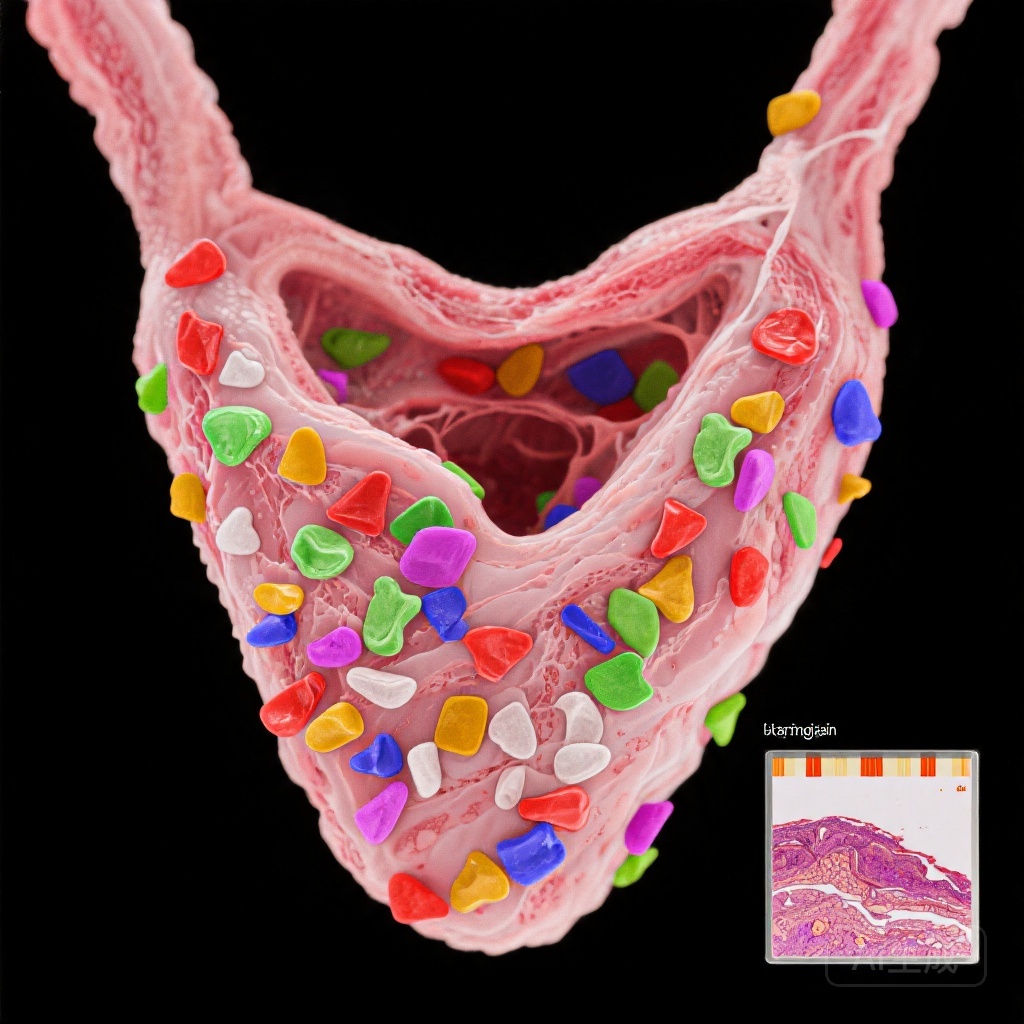
Microplastics in Human Thyroid: A Potential Trigger for Autoimmune Thyroiditis
Microplastics detected in thyroid tissues show higher concentrations in autoimmune thyroiditis patients, suggesting a potential environmental link to thyroid autoimmunity.