Highlights
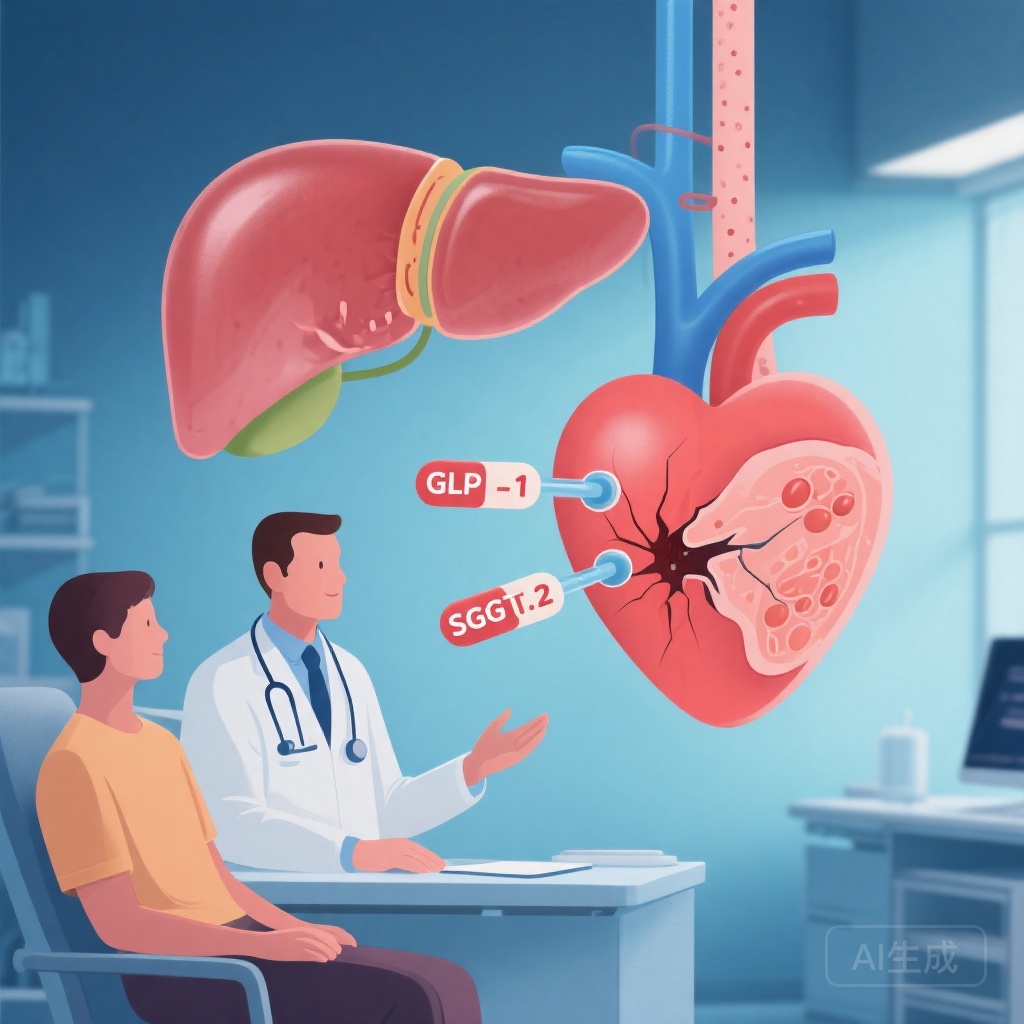
The combination of GLP-1 receptor agonists and SGLT-2 inhibitors reduces the risk of major adverse liver outcomes by 39% compared to GLP-1 receptor agonists alone and by 43% compared to SGLT-2 inhibitors alone. Additionally, the combination therapy lowers the risk of major adverse cardiovascular events by 35% and 20%, respectively.
Background
Metabolic dysfunction-associated steatotic liver disease (MASLD) is a growing global health concern, particularly among individuals with type 2 diabetes (T2D). Both GLP-1 receptor agonists and SGLT-2 inhibitors have shown individual benefits in managing MASLD and cardiovascular risks. However, the synergistic effects of these therapies remain underexplored.
Study Design
This target trial emulation study utilized data from U.S. Marketscan Databases to compare the outcomes of patients receiving combination therapy versus monotherapy. Propensity score-matched cohorts were constructed to ensure comparability. The study included 4606 patients on combination therapy compared to 18424 on GLP-1 receptor agonists alone, and 5368 on combination therapy compared to 21472 on SGLT-2 inhibitors alone.
Key Findings
The combination therapy demonstrated a significant reduction in major adverse liver outcomes, with hazard ratios of 0.61 (95% CI 0.49 to 0.77) compared to GLP-1 receptor agonists alone and 0.57 (95% CI 0.46 to 0.69) compared to SGLT-2 inhibitors alone. For major adverse cardiovascular events, the hazard ratios were 0.65 (95% CI 0.60 to 0.70) and 0.80 (95% CI 0.75 to 0.86), respectively.
Expert Commentary
The findings suggest that combining GLP-1 receptor agonists and SGLT-2 inhibitors may offer enhanced protection against liver and cardiovascular complications in patients with MASLD and T2D. However, the study’s observational nature and relatively short follow-up period (median 6.3 and 6.0 months) warrant cautious interpretation.
Conclusion
This study provides compelling evidence that the combination of GLP-1 receptor agonists and SGLT-2 inhibitors is associated with lower risks of major adverse liver and cardiovascular outcomes in patients with MASLD and T2D. Further randomized controlled trials are needed to confirm these findings and establish long-term efficacy and safety.
Funding
The study was supported by data from U.S. Marketscan Databases. ClinicalTrials.gov registration number was not provided.
References
Mao X, Fan H, Yuen MF, Cheung R, Seto WK, Nguyen MH. GLP-1 receptor agonist-SGLT-2 inhibitor combination and risk of major adverse liver and cardiovascular outcomes in adults with MASLD and type 2 diabetes. Hepatology (Baltimore, Md.). 2026-04-23. PMID: 42029657.