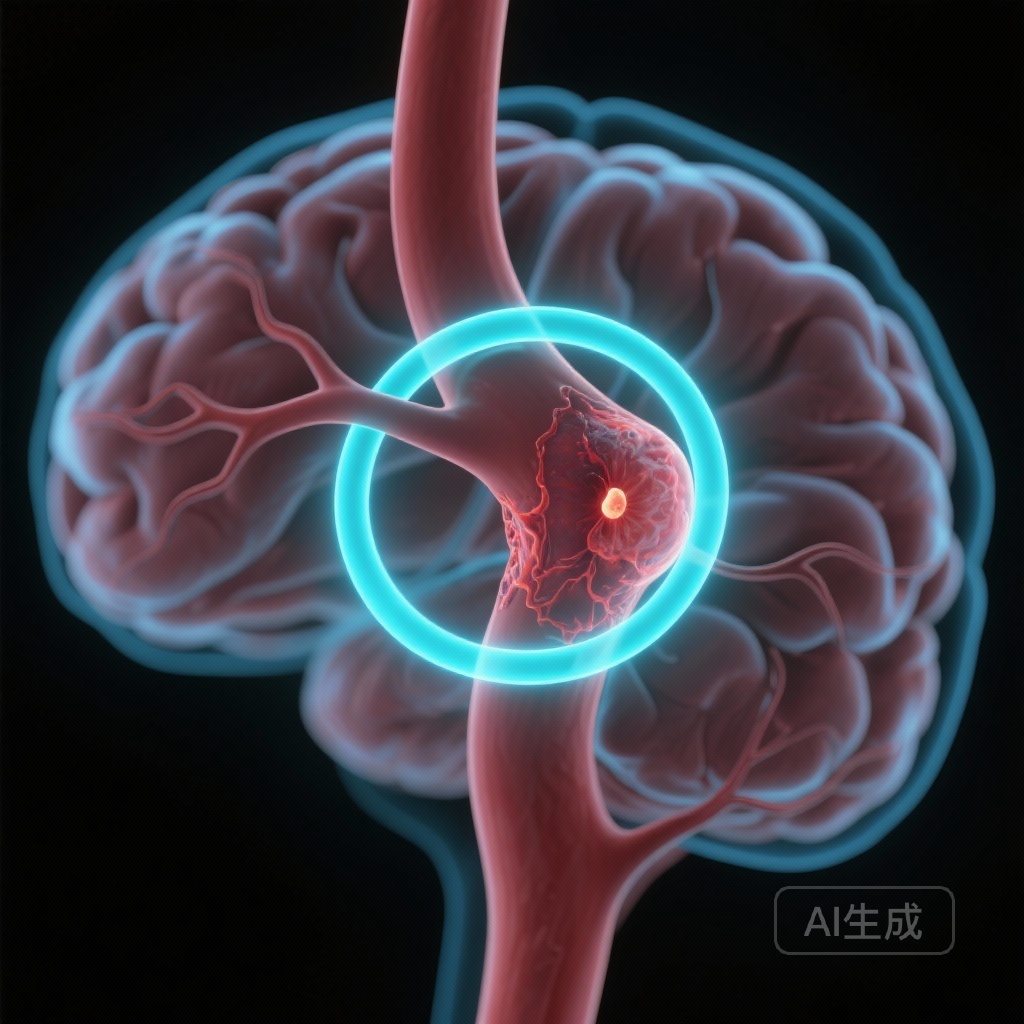
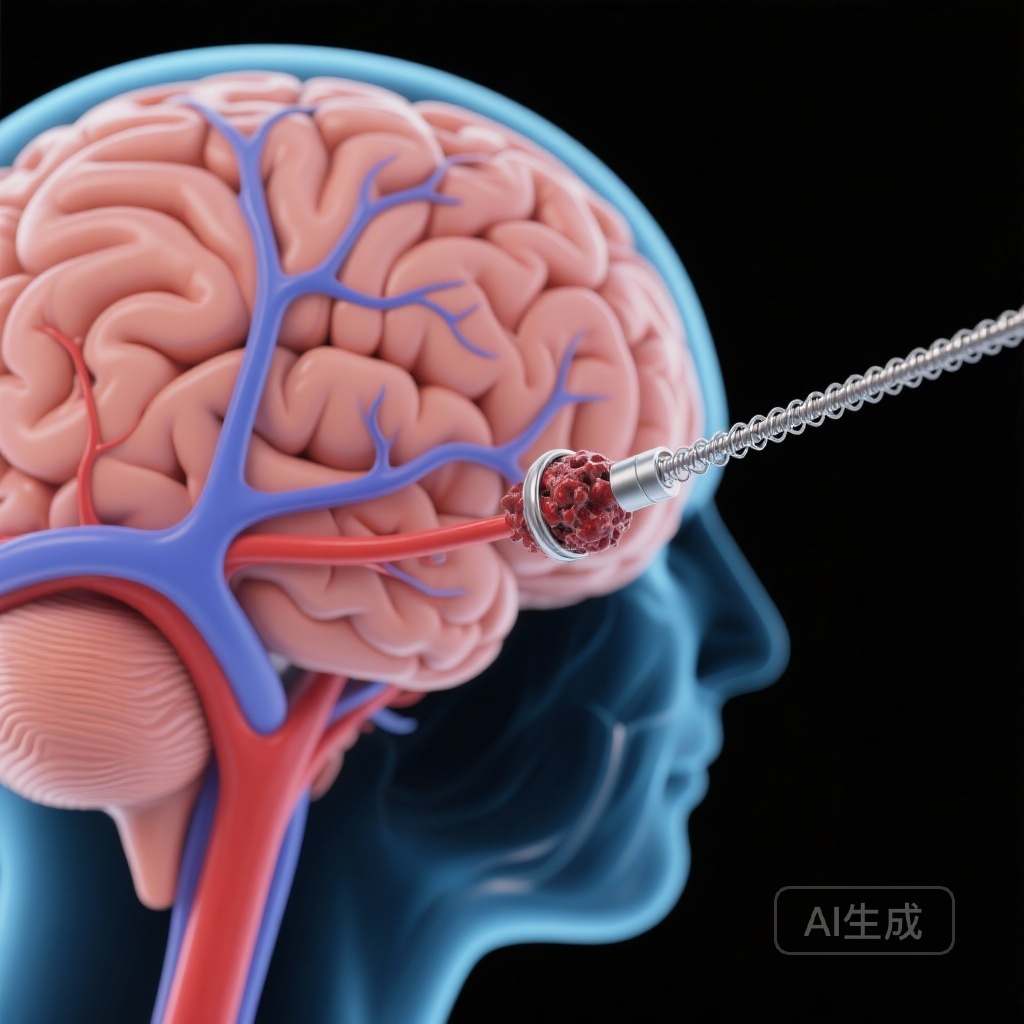
Circumferential Aneurysm Wall Enhancement Predicts a Three-Fold Increase in 4-Year Risk of Instability
A large-scale prospective study in JAMA Neurology identifies circumferential aneurysm wall enhancement (AWE) as a potent independent predictor of intracranial aneurysm growth or rupture, offering a critical radiological biomarker for clinical risk stratification.