Highlights
This feasibility randomized controlled trial investigated real-time functional magnetic resonance imaging (fMRI) neurofeedback as a novel intervention for poststroke reading deficits. Contingent neurofeedback targeting the left supramarginal gyrus produced reading-specific activation changes and significant comprehension improvements compared with noncontingent controls. The findings support the feasibility of neuromodulation approaches during early stroke recovery, though confirmation awaits the ongoing definitive randomized controlled trial.
Key findings include significant reading comprehension improvements in the contingent neurofeedback group (mean difference 9.75 points versus healthy controls; 11.42 points versus noncontingent stroke group), accompanied by training-related activation increases localized to the left supramarginal gyrus during nonword reading tasks.
Background: Addressing Reading Impairments in Post-Stroke Aphasia
Reading impairments represent a common and debilitating consequence of stroke-induced aphasia, substantially limiting patients’ participation in functional activities, vocational pursuits, and leisure engagements. Aphasia affects approximately 21–38% of acute stroke survivors, with reading deficits persisting in a significant proportion even after standard speech-language therapy. Traditional rehabilitation approaches, while beneficial for some patients, demonstrate limited generalization to untrained tasks and contexts, highlighting an unmet need for novel interventions that target residual neural networks directly.
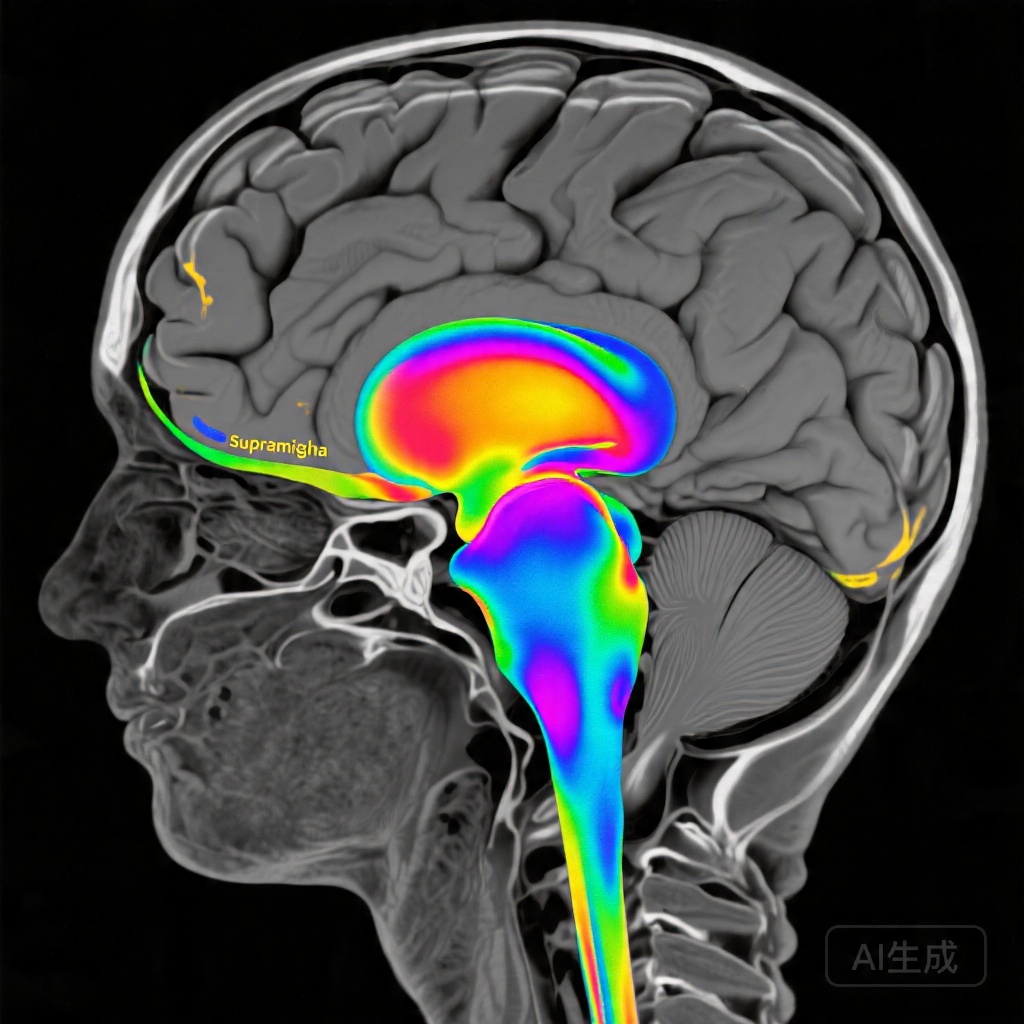
The emergence of real-time fMRI neurofeedback offers a promising neuromodulation strategy that enables patients to visualize and consciously modulate their own brain activity in specific regions implicated in language function. Unlike transcranial magnetic stimulation or transcranial direct current stimulation, fMRI neurofeedback provides direct hemodynamic-based feedback from the target region, potentially allowing for more precise targeting of task-specific neural circuits. The left supramarginal gyrus, a critical hub within the dorsal language pathway, plays an essential role in phonological processing and grapheme-to-phoneme conversion—skills fundamental to reading accuracy, particularly for novel or nonword stimuli.
The conceptual framework underlying this intervention draws from neuroplasticity principles suggesting that repeated, volitional modulation of task-relevant brain regions during early recovery periods may strengthen residual neural pathways and facilitate functional reorganization. However, the feasibility, safety, and preliminary efficacy of this approach in subacute stroke survivors with reading impairments had not been systematically evaluated prior to the present investigation.
Study Design
This single-center feasibility randomized controlled trial enrolled subacute left-hemisphere stroke survivors and age-matched healthy controls over a defined recruitment period. The study protocol comprised 3 weekly supervised fMRI neurofeedback sessions conducted within the imaging environment, complemented by 10 out-of-scanner practice sessions designed to reinforce in-scanner learning in ecological contexts.
Stroke participants were randomized using concealed allocation to one of two intervention arms: the contingent neurofeedback group (N=4) received feedback based on real-time hemodynamic activity from their own left supramarginal gyrus, whereas the noncontingent neurofeedback group (N=3) received shuffled feedback derived from another participant’s brain activity, thereby controlling for nonspecific effects of receiving visual feedback during scanning without any genuine contingent relationship between behavior and signal. Healthy controls (N=4) received contingent neurofeedback identical to the active stroke group and served as a normative reference for expected neuroplastic changes in intact neural systems.
Primary outcome measures included changes from baseline to postintervention (approximately 3 weeks following baseline assessment) in task-based brain activity during motor imagery, word reading, and nonword reading paradigms, as well as resting-state functional connectivity among motor, auditory, and language networks. Reading aloud accuracy served as a behavioral measure of decoding proficiency. Reading comprehension was evaluated as a secondary outcome using standardized assessment instruments.
Statistical analysis employed repeated-measures analysis of variance to examine group-by-session interaction effects, with planned contrasts comparing specific group differences in neurobiological and behavioral outcomes. Effect sizes were reported using partial eta-squared (η²) for omnibus tests and Cohen’s d-equivalent metrics for between-group comparisons, with 95% confidence intervals calculated for all primary effect estimates.
Study Population Characteristics
Participants were eligible if they had documented left-hemisphere ischemic or hemorrhagic stroke within the subacute phase (typically 1–6 months post-onset), confirmed aphasia affecting reading abilities, and adequate cognitive and auditory/visual function to participate in scanning procedures and neurofeedback training. Exclusion criteria included contraindications to MRI (e.g., ferromagnetic implants, claustrophobia), severe aphasia precluding task comprehension, active psychiatric comorbidities, or concurrent participation in other rehabilitation trials.
The healthy control cohort comprised age- and education-matched individuals without history of neurological or psychiatric illness. Demographic and clinical characteristics, including time since stroke, initial aphasia severity, and lesion characteristics, were documented to characterize the sample and inform interpretation of between-group differences.
Key Findings
Task-Based fMRI Results
Analysis of task-based functional imaging revealed striking differences in training-related activation patterns between the contingent and noncontingent neurofeedback groups. The contingent neurofeedback stroke group demonstrated significant activation increases localized to the left supramarginal gyrus during the postintervention scanning session (z=4.7; cluster-family-wise error corrected P=0.05), with expansion of activation within the broader reading network, particularly evident during nonword reading trials. This localization to a reading-specific region during phonologically demanding tasks suggests that the neurofeedback successfully reinforced activity within the intended target region during behaviorally relevant processing.
In contrast, activation increases in the noncontingent stroke group and healthy controls were more widespread and less reading-specific in their topography. Healthy controls showed engagement of additional executive and attentional control regions, potentially reflecting the greater cognitive demands of volitionally modulating an intact neural system, while the noncontingent stroke group demonstrated more diffuse patterns that may represent compensatory recruitment of alternative pathways without the specificity conferred by contingent feedback.
Resting-State Functional Connectivity
Resting-state fMRI analysis uncovered meaningful differences in network integration following the intervention period. The contingent neurofeedback groups (both stroke and healthy controls) exhibited greater integration among motor, auditory, and language networks, suggesting enhanced communication within distributed neural systems supporting sensorimotor integration and language processing. The noncontingent stroke group, conversely, demonstrated more disorganized connectivity patterns, with significant between-group differences confirmed by permutation testing (P=0.01) and small but meaningful effect sizes for connectivity changes (Δr=-0.1 to 0.1).
These findings align with theoretical models emphasizing that effective neuromodulation should enhance the coherence and functional integration of task-relevant networks rather than simply increasing regional activation in isolation. The specificity of connectivity changes to the contingent feedback conditions supports the mechanistic importance of the contingent relationship between brain activity and feedback presentation.
Behavioral Outcomes
Despite the promising neurobiological findings, no significant changes were observed in reading aloud accuracy across any of the groups. This dissociation between neural and behavioral outcomes at the level of single-word decoding may reflect the relatively short intervention duration (approximately 3 weeks), the primacy of neuroplastic changes preceding measurable behavioral gains, or the relative insensitivity of oral reading measures to subtle improvements in phonological processing efficiency.
However, a significant group-by-session interaction was identified for reading comprehension scores on the Reading Comprehension Battery for Aphasia, second edition (RCBA-2) (F[2, 8]=8.00; P<0.05; η²=0.67), indicating that the trajectory of change over the intervention period differed meaningfully between groups. Critically, the contingent neurofeedback stroke group demonstrated greater improvement compared with healthy controls (mean difference in RCBA-2 change=9.75 points; 95% CI, 1.99–17.51; t[6]=3.07; P<0.05) and compared with the noncontingent neurofeedback stroke group (mean difference=11.42 points; 95% CI, 1.12–21.71; t[5]=2.85; P<0.05).
The observation that stroke patients with reading impairments showed greater comprehension improvements than healthy controls with intact reading abilities is notable and suggests that the contingent neurofeedback intervention may have specifically targeted deficit-resolution mechanisms in the impaired system rather than simply enhancing already-potent reading networks.
Expert Commentary
The present findings contribute valuable preliminary evidence to the emerging field of fMRI neurofeedback as a neuromodulation strategy for language rehabilitation. Several methodological strengths warrant acknowledgment, including the randomized controlled design with an active sham condition, the use of task-specific and resting-state neuroimaging outcomes, and the inclusion of a healthy control group for normative reference. The emphasis on early recovery stages aligns with theoretical models suggesting that neuroplasticity windows may be particularly responsive to targeted stimulation during this period.
However, several limitations qualify interpretation of these preliminary results. The very small sample sizes (4 participants per contingent group, 3 in the noncontingent stroke group) preclude definitive conclusions about efficacy and limit statistical power for detecting smaller effect sizes. The absence of long-term follow-up assessments means that the durability of observed improvements remains unknown. Additionally, the single-center design with specialized neuroimaging expertise may constrain generalizability to broader clinical settings with different resource availabilities.
The mechanistic pathway through which contingent neurofeedback improves reading comprehension without corresponding changes in reading aloud raises important questions for future investigation. One possibility is that the intervention preferentially targets top-down semantic integration processes underlying comprehension while leaving bottom-up decoding efficiency relatively unchanged. Alternatively, the RCBA-2 may capture improvements in higher-order reading strategies or attentional allocation that do not manifest as changes in oral reading accuracy. Elucidating the cognitive and neural mechanisms mediating these improvements will be essential for optimizing the intervention and identifying optimal target populations.
Implications for Clinical Practice and Research
While these findings are preliminary, they suggest that real-time fMRI neurofeedback targeting the left supramarginal gyrus represents a technically feasible and potentially effective approach for addressing reading comprehension deficits in subacute stroke survivors with aphasia. The large effect size for the group-by-session interaction (η²=0.67) indicates substantial between-group differences in response trajectories, and the specificity of effects to the contingent condition supports the mechanistic importance of the feedback contingency itself rather than nonspecific factors such as scanner exposure or practice effects.
The ongoing definitive randomized controlled trial registered at ClinicalTrials.gov (NCT04875936) will be essential for confirming these preliminary observations in adequately powered samples. Future directions include investigation of optimal dosing parameters (number and spacing of sessions), identification of patient-specific predictors of response (e.g., lesion location, baseline network integrity), comparison with established speech-language therapy approaches, and long-term follow-up to assess durability of benefits.
From a translational perspective, the technical infrastructure required for real-time fMRI neurofeedback currently limits immediate clinical applicability to specialized research centers. However, as the technology matures and becomes more accessible, this approach may eventually complement or enhance traditional rehabilitation strategies by directly engaging residual neural circuits in a closed-loop paradigm that empowers patients to become active agents in their own recovery.
Conclusion
This feasibility randomized controlled trial provides promising preliminary evidence that contingent real-time fMRI neurofeedback targeting the left supramarginal gyrus can induce reading-specific neuroplastic changes and improve comprehension outcomes in subacute stroke survivors with aphasia. The specificity of findings to the contingent feedback condition, combined with observed changes in resting-state network integration, supports further investigation of this neuromodulation approach as a novel rehabilitation strategy.
Key takeaways include the feasibility of implementing fMRI neurofeedback in the subacute stroke population, the potential for neurofeedback to produce reading-specific rather than generalized neural changes, and the encouraging preliminary evidence for comprehension improvements that exceed those observed in healthy controls or noncontingent feedback conditions. Confirmation of these effects in adequately powered trials will be critical before considering clinical implementation.
Significant research gaps remain regarding optimal intervention parameters, patient selection criteria, mechanistic mediators of treatment response, and long-term outcomes. The ongoing randomized controlled trial (NCT04875936) will provide more definitive evidence to guide future development of this promising therapeutic approach.
Funding and Registration
This study was registered at ClinicalTrials.gov under Unique Identifier: NCT04875936. Complete funding disclosures and additional study documentation are available through the trial registration record.
References
Boukrina O, Yamin A, Yue GH, Kong Y, Koush Y. Feasibility Randomized Controlled Trial of Real-Time fMRI Neurofeedback for Reading Rehabilitation in Aphasia. Stroke. 2026-04-10. PMID: 41958417.