Patient Information
Between June 21, 2021, and December 5, 2022, six pregnant women were enrolled in the CuRe trial at the University of California, Davis (UC Davis) School of Medicine. The participants were carrying fetuses diagnosed with myelomeningocele (MMC). The maternal-fetal pairs were enrolled in a staggered manner to ensure safety monitoring. Eligibility criteria included a gestational age between 19 and 26 weeks, a normal fetal karyotype, and a myelomeningocele defect located between the T1 and S1 vertebrae. Additionally, all fetuses demonstrated hindbrain herniation on MRI, a common complication of MMC.
Diagnosis
The primary diagnosis for all six cases was fetal myelomeningocele, the most severe form of spina bifida. Diagnosis was confirmed using high-resolution ultrasound and fetal MRI. Key diagnostic markers included the identification of the neural tube defect, the level of the spinal lesion (ranging from T1 to S1), and the presence of the Chiari II malformation, specifically hindbrain herniation. The reasoning for enrollment was based on the potential for cellular therapy to augment standard surgical repair and improve motor outcomes that are often limited even with traditional in-utero surgery.
Differential Diagnosis
In the prenatal setting, myelomeningocele must be differentiated from other forms of spinal dysraphism and neural tube defects. These include:
- Myeloschisis: A similar open neural tube defect where the spinal cord is exposed without a covering sac.
- Lipomyelomeningocele: A closed neural tube defect involving a fatty mass.
- Sacrococcygeal teratoma: A tumor at the base of the coccyx which can sometimes mimic the appearance of a neural sac.
For this trial, diagnosis was refined through genetic testing (karyotyping) and detailed imaging to ensure the defect met specific anatomical criteria for the experimental cellular treatment.
Treatment and Management
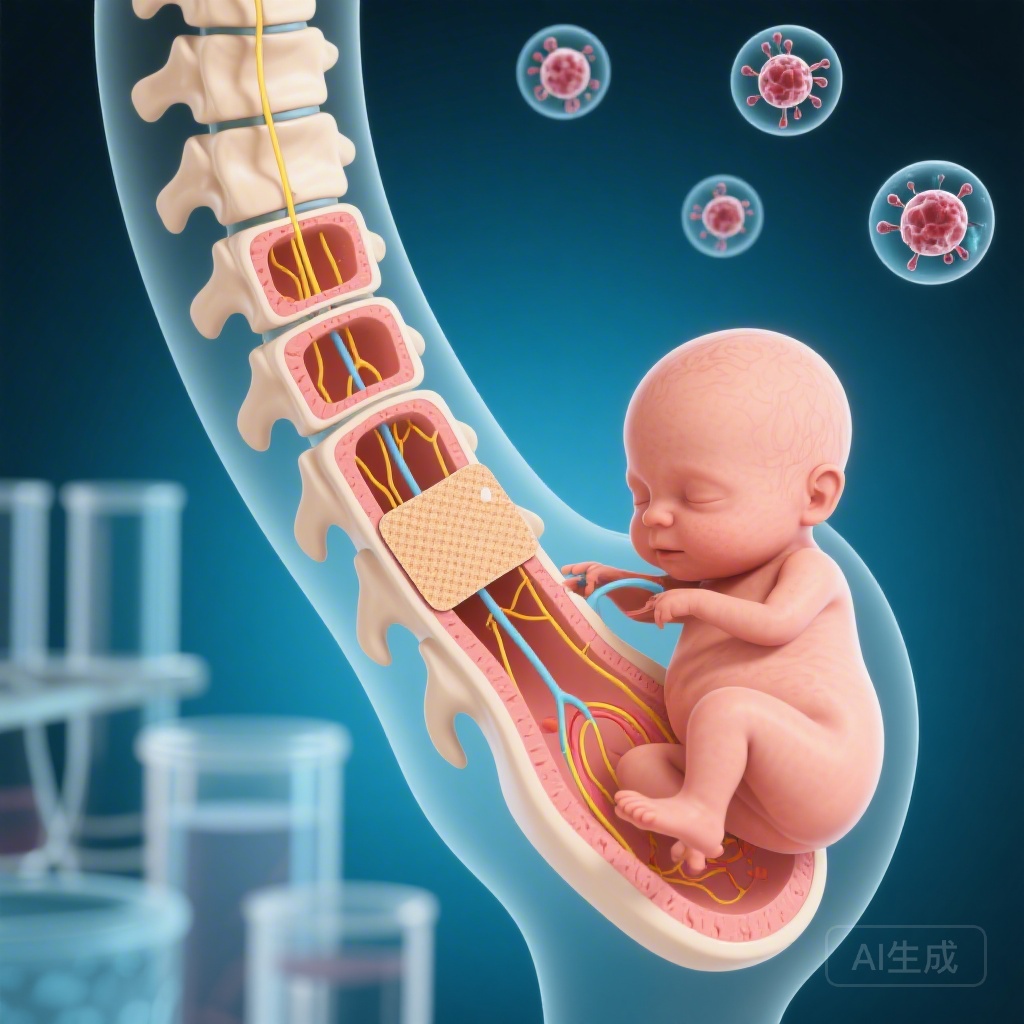
The intervention consisted of a novel in-utero repair technique. Standard fetal surgery was augmented with a single dose of topically applied allogeneic human placenta-derived mesenchymal stem cells (PMSCs). These cells were seeded onto a Cook Biodesign Dural Graft (an extracellular matrix).
The PMSCs were harvested from donated placentas, with strict adherence to identity, sterility, and viability protocols 72 hours prior to the surgery. During the open fetal surgery (conducted between 24+5 and 25+5 weeks’ gestation), the neurosurgical repair of the MMC defect was performed, and the stem cell-seeded graft was placed over the spinal cord. The goal was to provide a neuroprotective environment to rescue neurological function that is typically lost during gestation due to exposure of the spinal cord to amniotic fluid.
Outcome and Prognosis
The median gestational age at delivery was 34+5 weeks (range 33+2 to 36+6 weeks) via cesarean section. Primary safety endpoints were successfully met:
- All infants showed an intact repair site at birth.
- No evidence of cerebrospinal fluid (CSF) leak or infection was found.
- There was no evidence of abnormal tissue growth or tumor formation.
- Post-treatment MRIs revealed a significant reversal of hindbrain herniation in all subjects.
- No cell-mediated adverse events occurred in the mothers or the newborns.
While the phase 1 study focused on safety, the absence of complications and the successful anatomical repair suggest a favorable prognosis for the subsequent phase of the trial.
Discussion
The Management of Myelomeningocele Study (MOMS) trial previously proved that in-utero surgery reduces the need for shunting, yet many children remain unable to walk independently. The CuRe trial introduces regenerative medicine into the surgical field to address this functional gap.
This first-in-human application of allogeneic PMSCs is significant because it utilizes the neuroprotective properties of stem cells to mitigate the “second hit” of MMC—the chemical and mechanical damage to the exposed spinal cord in utero. The findings from these six patients demonstrate the feasibility of combining complex fetal surgery with living cellular products. The reversal of hindbrain herniation is a particularly encouraging sign of successful surgical and biological intervention. Given the safety profile established here, the study has been approved to proceed to a phase 1/2a trial involving a larger cohort (35 patients) to further evaluate efficacy and motor function improvements.
References
Farmer DL, Kumar P, Reynolds E, et al. Feasibility and safety of cellular therapy for in-utero repair of myelomeningocele (CuRe Trial): a first-in-human, phase 1, single-arm study. Lancet. 2026 Feb 28;407(10531):867-875. doi: 10.1016/S0140-6736(25)02466-3. PMID: 41763744.