Introduction
The management of chronic spontaneous urticaria (CSU) has long been a challenge for clinicians, particularly when standard-of-care treatments fail to provide adequate relief. Characterized by the sudden appearance of itchy hives, angioedema, or both for six weeks or longer, CSU significantly impairs quality of life, sleep, and mental health. While second-generation H1-antihistamines (H1-AH) are the first-line therapy, up to 50% of patients remain symptomatic even with fourfold doses. For these refractory patients, the therapeutic landscape has been relatively narrow. However, recent findings from the LIBERTY-CSU CUPID Phase 3 clinical trial program, specifically the CUPID-C study and pooled analyses, suggest that dupilumab—a monoclonal antibody targeting the IL-4/IL-13 signaling pathway—offers a significant breakthrough for patients who are anti-IgE naive and uncontrolled on antihistamines.
Highlights
The following points summarize the primary takeaways from the LIBERTY-CSU CUPID-C trial and the combined analysis:
Efficacy in Refractory CSU
Dupilumab significantly reduced itch and hive severity in patients who remained symptomatic despite standard or high-dose antihistamine therapy.
Confirmation of Previous Findings
The CUPID-C trial successfully replicated the positive outcomes of the earlier CUPID-A trial, meeting all primary and key secondary endpoints as required for regulatory consideration.
Targeted Type 2 Inhibition
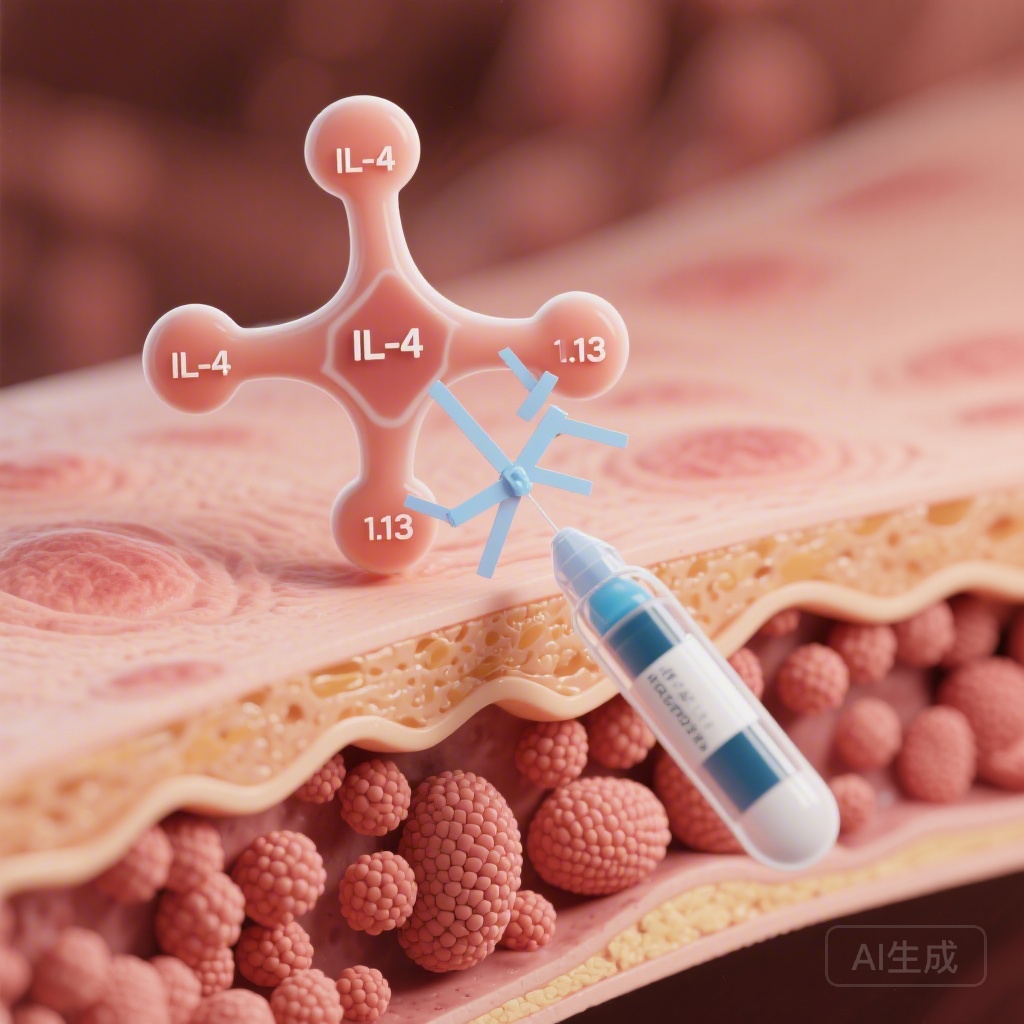
The study reinforces the role of type 2 inflammation (mediated by IL-4 and IL-13) in the pathogenesis of CSU, expanding the clinical utility of dupilumab beyond asthma and atopic dermatitis.
Favorable Safety Profile
Safety outcomes were consistent with the established profile of dupilumab, with adverse event rates comparable to the placebo group.
Background: The Burden and Pathophysiology of CSU
CSU is more than just a skin condition; it is a systemic inflammatory disease with a profound psychosocial burden. The traditional understanding of CSU centers on the activation of mast cells and basophils, leading to the release of histamine and other inflammatory mediators. This activation is often driven by autoimmune mechanisms, including Type I autoimmunity (IgE-mediated) and Type IIb autoimmunity (IgG-mediated).Current international guidelines, such as those from the EAACI/GA²LEN/EuroGuiDerm/APAAACI, recommend a stepwise approach starting with H1-AH. When these fail, omalizumab (an anti-IgE antibody) is typically the next step. However, access to omalizumab and varied patient responses leave a clinical gap. The exploration of dupilumab in this space is based on the observation that IL-4 and IL-13 contribute to IgE production and the recruitment and sensitization of inflammatory cells in the skin. By blocking the shared IL-4 receptor alpha (IL-4Rα) subunit, dupilumab interrupts the signaling of both cytokines, potentially stabilizing the inflammatory environment that leads to urticaria.
Study Design: The LIBERTY-CSU CUPID Program
The LIBERTY-CSU CUPID-C (NCT04180488) was a randomized, placebo-controlled, double-blind, 24-week phase 3 trial. It was designed as a replicate of the CUPID-A trial to satisfy US Food and Drug Administration (FDA) requirements for evidence of efficacy and safety.
Participant Demographics and Inclusion
The trial enrolled 151 participants across 10 countries. Inclusion criteria focused on patients aged 6 to 80 years with documented CSU that was inadequately controlled by H1-AH. Notably, all participants were naive to anti-IgE therapy (omalizumab). At baseline, over 50% of the cohort was using antihistamine doses higher than recommended, and nearly 60% had a baseline Urticaria Activity Score over 7 days (UAS7) of 28 or greater, indicating high disease activity.
Intervention and Endpoints
Participants were randomized to receive either dupilumab (300 mg every two weeks for adults, with weight-based dosing for children) or a matching placebo for 24 weeks. The primary endpoints were the change from baseline to week 24 in the Itch Severity Score over 7 days (ISS7) and the UAS7. These scores are validated tools for measuring patient-reported disease severity, with higher scores reflecting more intense itching and a higher number of hives.
Key Findings: Significant Reductions in Disease Activity
The results from CUPID-C and the pooled analysis with CUPID-A provide robust evidence for the clinical benefit of dupilumab.
CUPID-C Results
In the CUPID-C trial, dupilumab demonstrated statistically significant superiority over placebo. For the ISS7, the least squares (LS) mean change from baseline was -8.64 for the dupilumab group compared to -6.10 for the placebo group, representing a treatment difference of -2.54 (95% CI, -4.65 to -0.43; P = .02). Similarly, for the UAS7, the LS mean change was -15.86 for dupilumab versus -11.21 for placebo, a difference of -4.65 (95% CI, -8.65 to -0.65; P = .02).
Pooled Analysis (CUPID-A and CUPID-C)
When data from both trials (totaling 289 participants) were combined, the magnitude of the effect became even more evident. The pooled analysis showed a consistent and robust improvement in itch and hive severity across different geographic regions and age groups. This consistency suggests that the therapeutic effect of dupilumab in CSU is reliable and reproducible.
Secondary Endpoints
Beyond the primary scores, dupilumab-treated patients reported better quality of life and a higher likelihood of achieving complete or near-complete control of their symptoms compared to those on placebo. These patient-centered outcomes are critical in CSU management, where the subjective experience of itch is the primary driver of disability.
Safety and Tolerability
Safety is a paramount concern when introducing biological therapies. In the pooled analysis, 53.5% of patients in the dupilumab group reported treatment-emergent adverse events (TEAEs), compared to 55.9% in the placebo group. Most events were mild to moderate in severity. The most common TEAEs included injection site reactions and headache, which are consistent with the known safety profile of dupilumab in other indications like asthma and atopic dermatitis. There were no new or unexpected safety signals, affirming that dupilumab is a well-tolerated option for long-term management of CSU.
Expert Commentary: Mechanistic Insights and Clinical Impact
The success of the CUPID program has significant implications for our understanding of CSU. Historically, CSU was viewed primarily through the lens of histamine and IgE. However, the efficacy of an IL-4/IL-13 blocker highlights the complexity of the disease’s immune landscape. By reducing the Th2-driven environment, dupilumab may not only lower IgE levels over time but also decrease the sensitivity of mast cells to various triggers.From a clinical perspective, these findings suggest that dupilumab could serve as a valuable alternative or addition to the current treatment algorithm. While omalizumab remains a cornerstone of therapy, having a second biologic with a different mechanism of action is essential for personalized medicine. For instance, patients who have comorbid conditions like asthma or atopic dermatitis—both of which are dupilumab-responsive—may particularly benefit from this approach.One limitation noted by experts is the exclusion of patients who had previously failed omalizumab in these specific trial analyses (CUPID-A and C). While a separate trial (CUPID-B) investigated that specific population, the results discussed here are strictly applicable to those who are anti-IgE naive. Future research should continue to explore biomarkers that can predict which patients will respond best to IL-4/IL-13 inhibition versus anti-IgE therapy.
Conclusion
The LIBERTY-CSU CUPID-C trial, along with the pooled data from CUPID-A, provides high-level evidence that dupilumab is an effective and safe treatment for patients with chronic spontaneous urticaria that is not controlled by antihistamines. By significantly reducing the burden of itch and hives, dupilumab addresses a major unmet need in the dermatology and allergy community. As regulatory bodies review these data, clinicians can look forward to a potential expansion of the therapeutic toolkit, offering new hope to patients living with the daily challenges of CSU.
Funding and Clinical Registration
The LIBERTY-CSU clinical trial program was funded by Sanofi and Regeneron Pharmaceuticals. The trial is registered at ClinicalTrials.gov under the identifier NCT04180488.
References
Casale TB, Saini SS, Ben-Shoshan M, Giménez-Arnau AM, Bernstein JA, Hayama K, Amin N, Robinson LB, Bauer D, Dakin P, Laws E, Radin A, Makhija M. Dupilumab in Patients With Chronic Spontaneous Urticaria: Phase 3 LIBERTY-CSU CUPID Randomized Clinical Trials. JAMA Dermatol. 2026 Feb 18:e256023. doi: 10.1001/jamadermatol.2025.6023. Epub ahead of print. PMID: 41706458; PMCID: PMC12917742.