Highlights
- BMPR1B and TAZ/TEAD expression levels are significantly elevated in relapsed AML patients and cells resistant to cytarabine or venetoclax.
- Bone marrow physical changes, including extracellular matrix stiffening and increased intramedullary pressure, drive leukemic persistence.
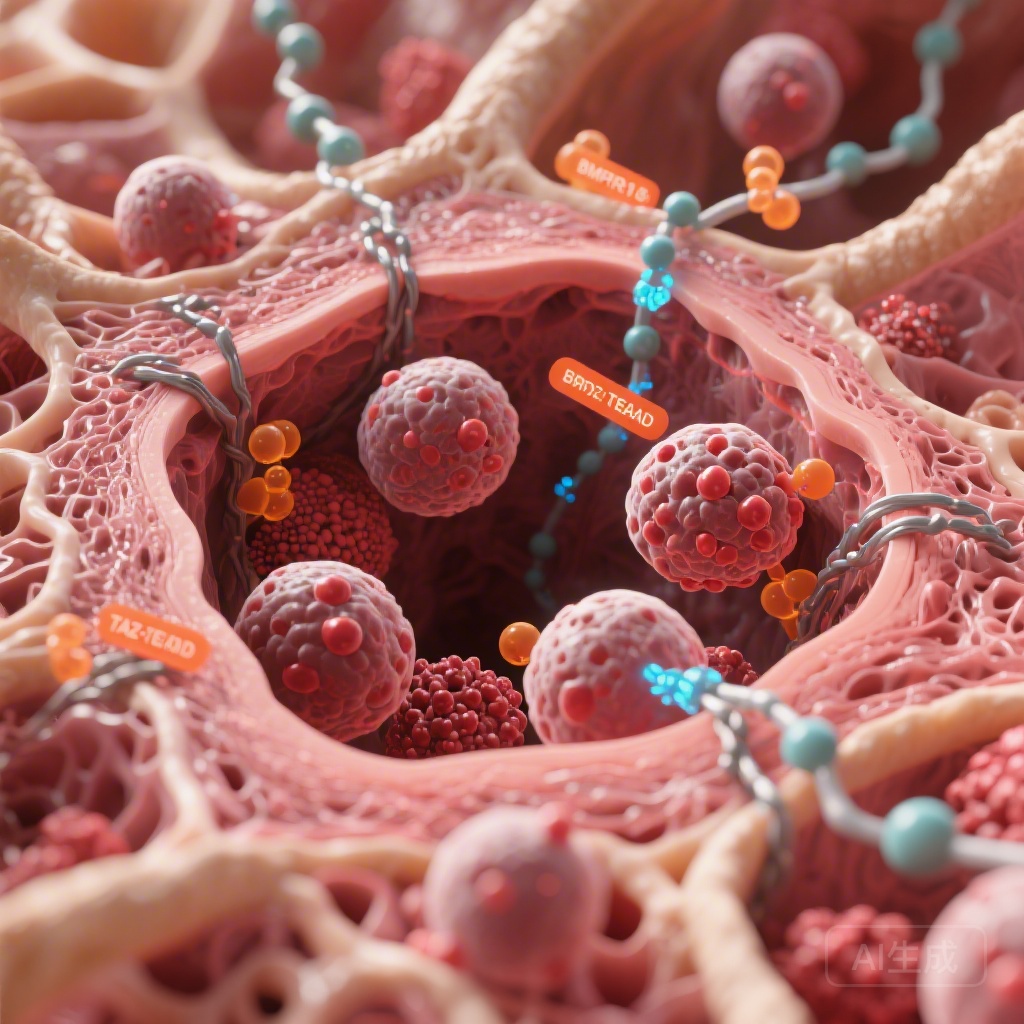
- Mesenchymal stem cells (MSCs) under high confinement secrete BMP4, which specifically activates chemoresistant AML cells.
- Combined inhibition of BMPR1B or TAZ/TEAD with cytarabine effectively clears AML primary cells in 3D human bone marrow models.
Introduction: The Biomechanical Landscape of AML
For decades, the management of acute myeloid leukemia (AML) has focused almost exclusively on the genetic and epigenetic landscape of leukemic blasts. Despite the introduction of targeted therapies like FLT3 inhibitors and venetoclax, the 5-year overall survival rate remains a sobering 20%. This clinical plateau suggests that our understanding of chemoresistance is incomplete. A growing body of evidence now points toward the bone marrow (BM) microenvironment—not just as a passive reservoir for cells, but as an active physical participant in disease progression.
As leukemic blasts undergo clonal expansion, they fundamentally alter the physical properties of the bone marrow. This “biomechanical remodeling” includes increased extracellular matrix (ECM) stiffening, a rise in intramedullary pressure, and a significant reduction in the physical space available for resident cells. While these changes have been observed in solid tumors for years, their role in the survival of hematological malignancies is only now being elucidated. The study by Barral et al. (2026) provides a critical breakthrough, identifying mechanotransduction—the process by which cells convert mechanical stimuli into chemical signals—as a primary driver of chemoresistance in AML.
Study Design and Methodological Framework
The researchers employed a multi-faceted approach to investigate the role of mechanical stress in AML. The study utilized primary AML samples collected at diagnosis and relapse, as well as cell lines specifically engineered for resistance to standard-of-care agents, cytarabine (Ara-C) and venetoclax.
To simulate the physical constraints of the leukemic bone marrow, the team developed a 3D human bone marrow-like model. This platform allowed for the precise manipulation of matrix stiffness and cell confinement, providing a more physiologically relevant environment than traditional 2D cultures. The study focused on two key mechanotransduction pathways: the Bone Morphogenetic Protein (BMP) pathway and the Hippo pathway effectors, TAZ and YAP. Through transcriptomic analysis and protein quantification, the authors sought to determine how these pathways respond to the mechanical pressures of the leukemic niche.
Key Findings: The BMPR1B and TAZ/TEAD Axis
The Rise of BMPR1B and TAZ in Resistance
The analysis revealed a striking correlation between mechanotransduction signaling and clinical outcomes. Patients who relapsed after chemotherapy showed significantly higher levels of BMPR1B (a BMP receptor) and TAZ, along with its transcriptional partner TEAD, compared to their status at initial diagnosis. Intriguingly, while TAZ was upregulated, its paralog YAP—often considered the central mechanosensor in other tissues—did not show a similar increase. This suggests a specific, non-redundant role for TAZ in the context of the leukemic bone marrow.
This pattern was mirrored in vitro. AML cell lines resistant to cytarabine and venetoclax exhibited a constitutive activation of the TAZ/TEAD complex. These resistant cells were not just genetically different; they were mechanically distinct. They demonstrated improved adhesion to microenvironmental components like fibronectin and laminin, and an increased intrinsic deformability, allowing them to navigate and survive within the high-pressure, confined spaces of a crowded bone marrow.
Paracrine Signaling: The MSC Connection
The study also highlighted a critical cross-talk between leukemic cells and resident mesenchymal stem cells (MSCs). Under conditions of high physical confinement—mimicking the overcrowding of a blast-filled marrow—MSCs were found to express elevated levels of BMP4. This MSC-derived BMP4 acted as a paracrine signal that specifically activated BMPR1B on the surface of chemoresistant AML cells. This interaction appears to create a protective “niche effect,” where the physical stress of the disease triggers a survival signal that shields the blasts from chemotherapy-induced apoptosis.
Targeting the Mechanome to Sensitize Blasts
Perhaps the most clinically significant finding was the effect of pharmacological inhibition. Using the 3D human bone marrow model, the researchers demonstrated that targeting either BMPR1B or the TAZ/TEAD complex could break the cycle of resistance. When these mechanotransduction inhibitors were combined with cytarabine, there was a synergistic impairment of AML primary cell persistence. By blocking the cells’ ability to sense and respond to the stiffened, high-pressure environment, the treatment effectively “unmasked” the leukemic cells, making them vulnerable to conventional cytotoxic therapy once again.
Expert Commentary: Mechanistic Insights and Clinical Implications
The findings by Barral et al. represent a paradigm shift in how we approach refractory AML. Traditionally, we have viewed the bone marrow as a liquid compartment, but this research forces us to treat it more like a solid tumor microenvironment where physical forces dictate cellular fate.
The specific involvement of TAZ over YAP is a particularly interesting biological nuance. In many epithelial cancers, YAP and TAZ are often co-activated, but the leukemic niche may place unique mechanical demands on cells that favor the TAZ-TEAD transcriptional program. This specificity offers a narrower, and perhaps more effective, therapeutic window for drug development.
However, there are limitations to consider. While the 3D BM-like model is a significant step forward, it cannot fully replicate the systemic complexity of a human patient, including the immune system’s role in the niche. Furthermore, the intramedullary pressure in AML is dynamic; understanding how these pathways fluctuate during the various stages of induction and consolidation therapy will be vital for the timing of mechanotransduction-targeted interventions.
From a health policy and clinical trial perspective, these results suggest that we should begin incorporating biomechanical markers—perhaps through advanced imaging or specialized biopsy analysis—into our risk stratification for AML. If a patient presents with a particularly stiff or high-pressure marrow, they may be prime candidates for early intervention with BMPR1B or TAZ inhibitors.
Conclusion: A New Pillar of AML Therapy
The identification of BMPR1B and TAZ/TEAD as mediators of chemoresistance opens a new frontier in hematology-oncology. By addressing the physical reality of the leukemic bone marrow, we may finally be able to overcome the environmental mediated drug resistance that has long plagued AML treatment. Future therapeutic strategies that combine traditional cytostatics with mechanotransduction inhibitors hold the promise of deeper remissions and lower relapse rates for patients who currently have few options. As we move toward 2026 and beyond, the integration of biophysics into clinical oncology will likely become a cornerstone of precision medicine.
References
- Barral L, Lespinasse N, Martin Cardozo C, et al. Targeting BMP and TAZ/TEAD mechanotransduction pathways impairs acute myeloid leukemia chemoresistance. Leukemia. 2026-03-18. PMID: 41851473.
- Lampi MC, Reinhart-King CA. Targeting extracellular matrix stiffness to attenuate disease progression. Sci Transl Med. 2018;10(422).
- Passaro D, et al. Increased Bone Marrow Pressure Drives Hematopoietic Stem Cell Loss and Myeloid Neoplasia. Cell Stem Cell. 2021.
- Dupont S, et al. Role of YAP/TAZ in mechanotransduction. Nature. 2011;474(7350):179-183.