Highlights
- Anti-BCMA bispecific antibodies (bsAbs) cause more severe and frequent infections in multiple myeloma (MM) patients compared to anti-GPRC5D bsAbs.
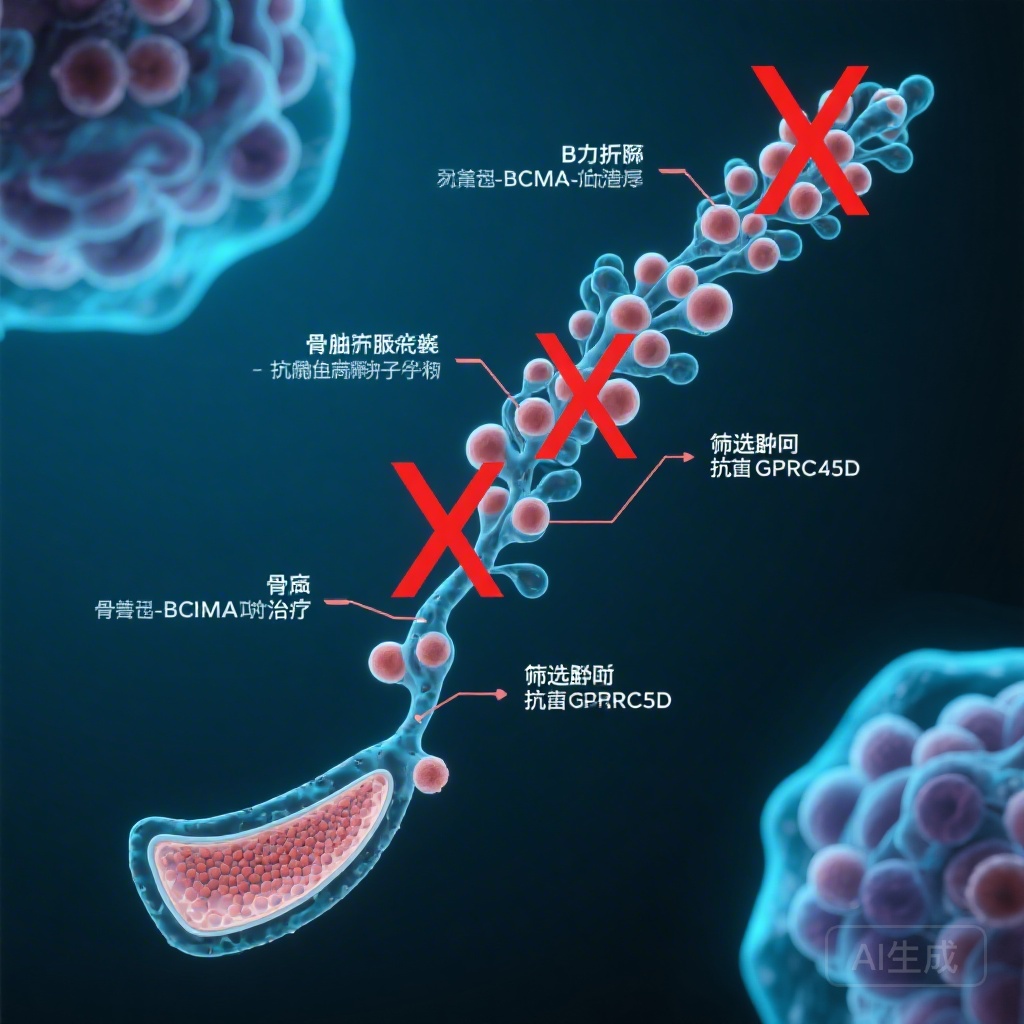
- The mechanism involves the unexpected expression of BCMA on early B-cell precursors, specifically small pre-B cells, leading to their depletion.
- In contrast, GPRC5D expression is highly restricted to malignant and normal plasma cells, sparing the earlier B-cell lineage.
- This study provides a biological rationale for individualized therapy selection based on a patient’s baseline infection risk and immune status.
Background: The Challenge of Infections in Modern Myeloma Therapy
The therapeutic landscape for relapsed/refractory multiple myeloma (RRMM) has been revolutionized by the introduction of T-cell redirecting therapies, particularly bispecific antibodies (bsAbs). These molecules bridge T cells to tumor-associated antigens, inducing potent cytotoxic responses. The two primary targets currently utilized in clinical practice are B-cell maturation antigen (BCMA) and G protein-coupled receptor class C group 5 member D (GPRC5D).
While both targets have demonstrated remarkable efficacy, a significant clinical disparity has emerged regarding safety profiles: patients treated with anti-BCMA bsAbs (such as teclistamab or elranatamab) experience significantly higher rates of high-grade infections and hypogammaglobulinemia compared to those receiving anti-GPRC5D agents (such as talquetamab). Until recently, the biological basis for this difference remained poorly understood, often attributed simply to the ubiquitous presence of BCMA on mature plasma cells. However, the depth and duration of the resulting lymphopenia suggested a more profound impact on the B-cell compartment than previously recognized.
Study Design and Methodology
To investigate this phenomenon, Jelinek et al. conducted a comprehensive multi-platform analysis published in Blood. The study utilized a combination of high-resolution transcriptomics, advanced flow cytometry, and animal models to map the expression of BCMA and GPRC5D across the entire spectrum of B-cell development.
The researchers performed single-cell RNA sequencing (scRNA-seq) on bone marrow (BM) aspirates from 11 patients with MM and 8 healthy donors to determine the precise cellular localization of target antigens. This was complemented by next-generation flow cytometry (NGF) immune profiling of 62 patients with relapsed MM undergoing treatment with various bsAbs. Additionally, the team utilized the MIcγ1 mouse model, which lacks BCMA expression across all B-cell stages, as a negative control to confirm that the observed cellular depletion was indeed target-specific. The study primary endpoints focused on the quantification of B-cell subsets and the correlation of these changes with clinical infection events.
Key Findings: Broad vs. Selective Depletion
BCMA Expression Beyond Plasma Cells
The most striking discovery was that BCMA expression is not restricted to mature B cells and plasma cells. Single-cell RNA sequencing revealed that BCMA is surprisingly expressed in small pre-B cells within the B-cell precursor compartment. In contrast, GPRC5D expression was found to be strictly limited to malignant plasma cells and, to a lesser extent, normal plasma cells, with virtually no expression on earlier B-cell subsets.
Clinical Evidence of Immune Eradication
The NGF profiling of patients provided clinical confirmation of these transcriptomic findings. In patients treated with anti-BCMA bsAbs, mature bone marrow B cells were severely depleted, dropping from a median of 4.9% to 0% (P < .001). Furthermore, normal plasma cells were effectively eradicated, decreasing from 0.17% to less than 0.0002% (P < .001). Crucially, this depletion was not transient; it persisted throughout the duration of therapy.
When comparing the two types of bsAbs, the data showed that anti-BCMA therapy, but not anti-GPRC5D therapy, led to the near-total loss of immature and small pre-B cells. This suggests that anti-BCMA agents do not just kill the ‘end-product’ (plasma cells) but also destroy the ‘factory’ (pre-B cells) responsible for replenishing the B-cell pool.
Animal Model Validation
The use of the MIcγ1 mouse model further solidified these results. In this model, where BCMA is not expressed at any stage of the B-cell lineage, treatment with anti-BCMA agents resulted in no depletion of B-cell subsets. This confirms that the lymphopenia observed in humans is a direct ‘on-target, off-tumor’ effect of the antibody’s mechanism rather than a non-specific consequence of T-cell activation.
Expert Commentary and Clinical Implications
The findings by Jelinek et al. have profound implications for the clinical management of multiple myeloma. The identification of BCMA on pre-B cells explains why patients on these therapies are so uniquely vulnerable to opportunistic infections. Because the B-cell ontogeny is interrupted at such an early stage, the body cannot mount a de novo humoral response to new pathogens.
Individualized Treatment Selection
Clinicians must now consider a patient’s baseline immune status and infection history when choosing between BCMA and GPRC5D targets. For a patient with a history of recurrent pneumonia or existing severe hypogammaglobulinemia, a GPRC5D-targeted approach may offer a safer therapeutic window by preserving the early B-cell compartments. Conversely, the broad depletion seen with BCMA may be leveraged in other contexts, such as autoimmune diseases, where the eradication of autoreactive B-cell precursors is a therapeutic goal.
Management of Infection Risk
The study underscores the necessity of aggressive supportive care for patients on anti-BCMA bsAbs. This includes routine intravenous immunoglobulin (IVIG) replacement, stringent antimicrobial prophylaxis (e.g., against PJP and HSV/VZV), and a high index of suspicion for atypical infections. The fact that the B-cell factory is effectively shut down means that these patients are functionally akin to those with severe combined immunodeficiency during the treatment period.
Conclusion
In summary, this research provides the definitive mechanistic insight into the infection profiles of modern myeloma therapies. By demonstrating that anti-BCMA bsAbs target the B-cell lineage from the small pre-B stage onward, while anti-GPRC5D bsAbs remain selective for plasma cells, the study clarifies why the former carries a significantly higher burden of infectious morbidity. These results support a move toward more individualized immunotherapy strategies in multiple myeloma, balancing the unprecedented efficacy of these agents with the need to preserve essential immune function.
References
1. Jelinek T, Zihala D, Zabaleta A, et al. Selective B-cell subset depletion underlies increased infection risk in patients with MM treated with anti-BCMA vs anti-GPRC5D bsAbs. Blood. 2026;147(10):1070-1082. PMID: 41405507.
2. Moreau P, Garfall AL, van de Donk NWCJ, et al. Teclistamab in Relapsed or Refractory Multiple Myeloma. N Engl J Med. 2022;387(6):495-505.
3. Chari A, Minnema MC, Berdeja JG, et al. Talquetamab, a T-Cell-Redirecting GPRC5D Bispecific Antibody, in Relapsed or Refractory Multiple Myeloma. N Engl J Med. 2022;387(24):2232-2244.