Highlights
Mitochondrial Plasticity Preserved
Individuals with type 2 diabetes (T2D) exhibit a preserved capacity for mitochondrial structural remodeling in response to exercise, comparable to lean and obese glucose-tolerant individuals.
HIIT Drives Ultrastructural Adaptation
Eight weeks of high-intensity interval training (HIIT) induced a ~7% increase in mitochondrial cristae density across all study groups, with specific enhancements in type 2 muscle fibers.
Significant Expansion of Oxidative Surface Area
HIIT led to a staggering ~55% increase in total cristae surface area per muscle volume, a metric that integrates both mitochondrial abundance and internal architecture, far exceeding typical gains in mitochondrial volume alone.
The Clinical Context: Beyond Mitochondrial Volume
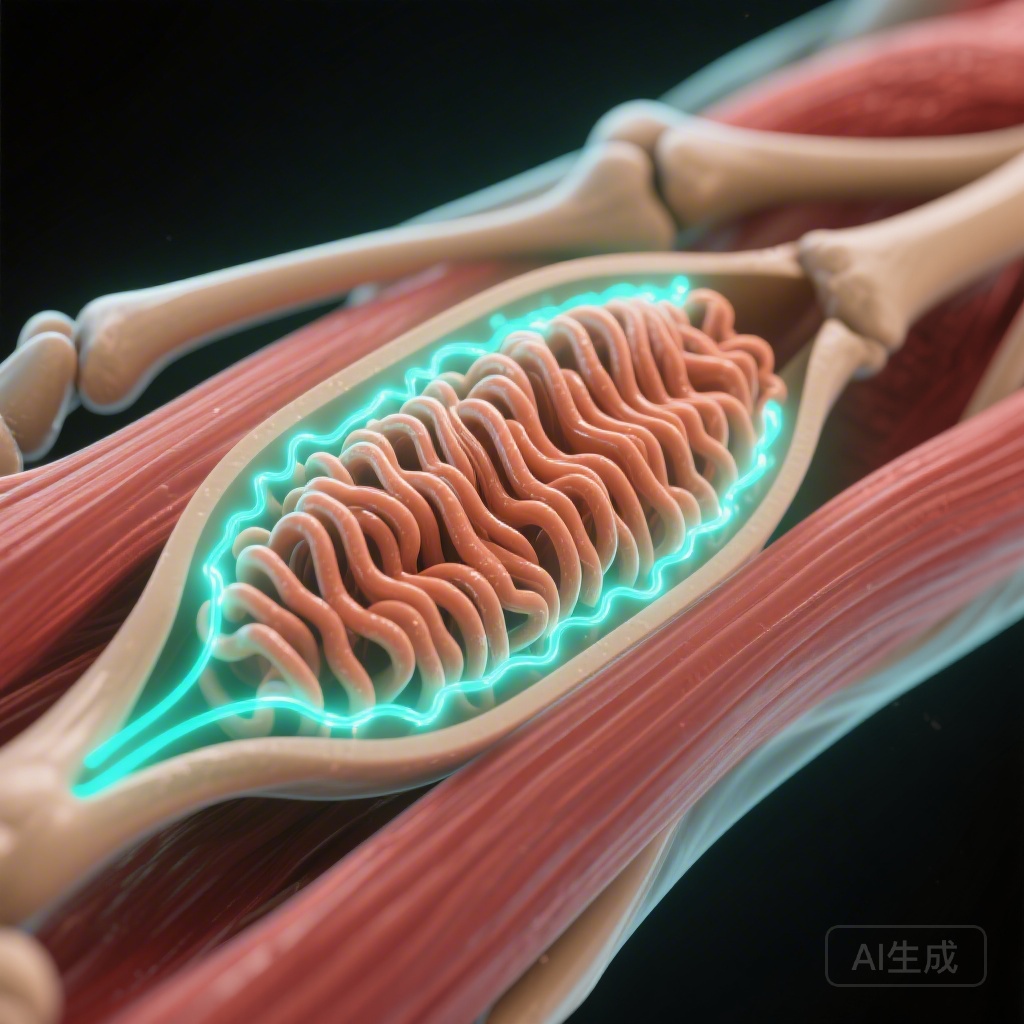
Skeletal muscle mitochondrial dysfunction has long been identified as a cornerstone in the pathophysiology of insulin resistance and type 2 diabetes. Historically, research has focused primarily on mitochondrial content—the total volume or number of mitochondria within a muscle cell. However, emerging evidence suggests that the quality and internal architecture of these organelles, specifically the density of the inner mitochondrial membrane folds known as cristae, may be a more critical determinant of oxidative capacity and metabolic health.
Cristae are the sites of the electron transport chain and ATP synthase. Therefore, the cristae surface area directly dictates the potential for oxidative phosphorylation (OXPHOS). While it is well-established that exercise increases mitochondrial volume, whether the internal ‘wiring’—the cristae density—is impaired in T2D or can be specifically modulated by exercise remained a subject of intense scientific debate. The study by de Almeida et al., published in Diabetologia, addresses this gap by providing a high-resolution analysis of mitochondrial ultrastructure in response to high-intensity interval training.
Study Design and Methodology
This non-randomized intervention study utilized a rigorous design to compare three distinct cohorts of men aged 40 to 65 years:
Study Populations
1. Individuals with type 2 diabetes (n=15).
2. Glucose-tolerant individuals with obesity (n=15).
3. Lean, healthy individuals (n=18).
The Intervention
Participants underwent an 8-week supervised HIIT program. The protocol combined rowing and cycling, ensuring a high-intensity stimulus designed to maximize metabolic stress and adaptation. The supervision ensured adherence and intensity consistency across the varied cohorts.
Ultrastructural Analysis
To assess mitochondrial changes at the nanoscopic level, muscle biopsies were taken from the vastus lateralis muscle before and after the intervention. The researchers employed transmission electron microscopy (TEM), the gold standard for visualizing cellular ultrastructure. To ensure statistical and stereological precision, the team analyzed a minimum of 49 mitochondrial profiles per sample, quantifying:
1. Mitochondrial cristae density (cristae surface area per mitochondrial volume).
2. Cristae surface area per muscle volume (a global measure of oxidative potential).
3. Compartment-specific changes (intermyofibrillar vs. subsarcolemmal mitochondria).
4. Fiber-type specific adaptations (Type 1 vs. Type 2 fibers).
Key Findings: A Triumph of Cellular Plasticity
The results of the study challenge several assumptions regarding mitochondrial ‘defects’ in diabetic muscle and highlight the potent therapeutic effect of HIIT.
Baseline Comparisons
Contrary to some previous hypotheses, the study found no significant differences in mitochondrial cristae density between the groups at baseline. This suggests that the individual mitochondria in a person with T2D are not inherently ‘broken’ or less ‘dense’ than those in a lean individual. However, the researchers did observe that participants with T2D had a lower total cristae surface area per muscle volume. This deficiency was driven by a lower overall mitochondrial volume density rather than a defect in the cristae themselves.
The Impact of HIIT on Cristae Density
Following the 8-week intervention, all groups showed a ~7% increase in cristae density. This indicates that exercise can change the internal structure of existing mitochondria, making them more ‘packed’ with the machinery for energy production. Interestingly, these adaptations were most pronounced in Type 2 (fast-twitch) muscle fibers and within the intermyofibrillar compartment—the mitochondria located between the contractile proteins, which are crucial for powering muscle contraction.
Massive Gains in Global Oxidative Surface Area
Perhaps the most clinically significant finding was the ~55% increase in total cristae surface area per muscle volume. This metric represents the cumulative effect of having more mitochondria (increased volume) and those mitochondria being more efficient (increased cristae density). The magnitude of this increase is notably higher than the typical 15-25% increase in mitochondrial volume density reported in previous HIIT studies, suggesting that the true ‘oxidative gain’ from exercise has been historically underestimated by only looking at volume.
Expert Commentary and Mechanistic Insights
The Plasticity of the Inner Membrane
This study reinforces the concept of mitochondrial plasticity. The inner mitochondrial membrane is not a static structure; it is a dynamic interface that responds to metabolic demand. The increase in cristae density likely involves the modulation of proteins such as OPA1 and the MICOS (Mitochondrial Contact Site and Cristae Organizing System) complex, which regulate cristae junction formation and membrane curvature.
Clinical Implications for T2D Management
For clinicians, these findings provide a strong biological rationale for prescribing HIIT to patients with type 2 diabetes. The fact that the ‘remodeling capacity’—the ability of the muscle to improve its mitochondrial quality—is not impaired by the diabetic state is highly encouraging. It suggests that even in the presence of chronic hyperglycemia and insulin resistance, the cellular machinery remains responsive to physiological stimulus.
Limitations and Future Directions
While the study provides high-resolution data, it was limited to male participants and utilized a non-randomized design. Future research should investigate whether these ultrastructural adaptations are equally present in women and how they correlate with long-term glycemic control (HbA1c) and insulin sensitivity. Additionally, the longevity of these structural changes after the cessation of training remains an open question.
Conclusion
In summary, skeletal muscle mitochondrial cristae density is not intrinsically reduced in men with type 2 diabetes compared to lean or obese counterparts. However, the total capacity for energy production is lower due to reduced mitochondrial volume. High-intensity interval training serves as a powerful corrective mechanism, not only by increasing the quantity of mitochondria but also by fundamentally remodeling their internal architecture. By increasing cristae density and total surface area, HIIT effectively expands the oxidative ‘workstation’ of the muscle, providing a robust pathway for improving metabolic health in patients with T2D.
References
1. de Almeida ME, Ørtenblad N, Platz AB, Petersen MH, Højlund K, Nielsen J. Mitochondrial cristae density is increased following high-intensity interval training in men with type 2 diabetes. Diabetologia. 2026. PMID: 41795037.
2. Nielsen J, Gejl KD, Hey-Mogensen M, et al. Plasticity of mitochondrial cristae density in human skeletal muscle with training and training cessation. Journal of Physiology. 2017.
3. Larsen S, Nielsen J, Hansen CN, et al. Biomarkers of mitochondrial content in skeletal muscle of healthy young human subjects. Journal of Physiology. 2012.