Highlights
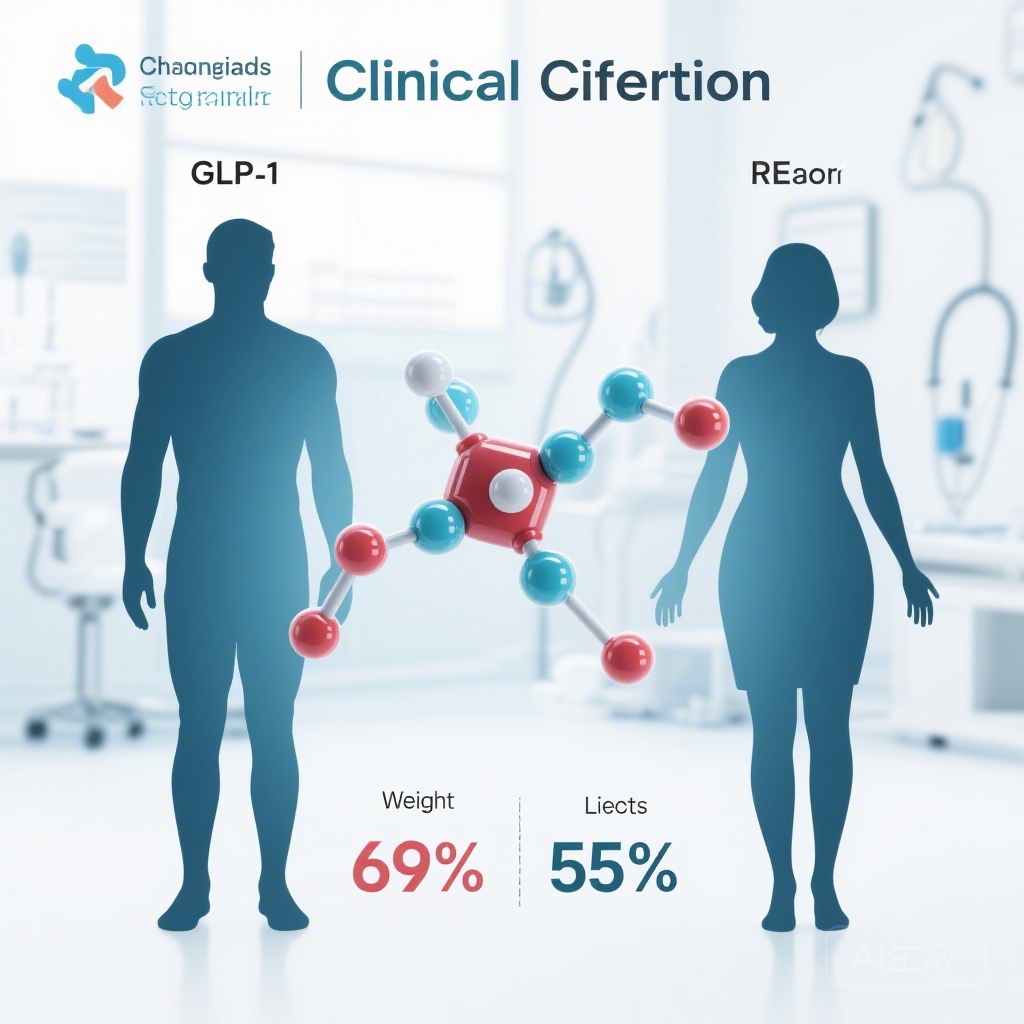
- Women experienced significantly greater weight loss (10.9%) compared to men (6.8%) when treated with GLP-1 receptor agonists (GLP-1 RAs).
- The efficacy of GLP-1 RAs remained consistent regardless of patient age, race, ethnicity, baseline BMI, or baseline HbA1c levels.
- The meta-analysis included 64 randomized clinical trials (RCTs), with semaglutide and dulaglutide being the most frequently evaluated agents.
- These findings suggest that while GLP-1 RAs are broadly effective, sex is a critical biological variable in determining the magnitude of treatment response.
Background: The Evolution of GLP-1 Receptor Agonists
The landscape of chronic disease management has been transformed by the emergence of glucagon-like peptide-1 receptor agonists (GLP-1 RAs). Originally developed for the treatment of type 2 diabetes, these agents—including semaglutide, liraglutide, and dulaglutide—have demonstrated profound effects on weight regulation and cardiovascular outcomes. As clinical use expands from specialized endocrinology clinics to primary care and obesity medicine, understanding the Heterogeneity of Treatment Effects (HTE) has become a priority for clinicians.While the overall efficacy of these agents is well-documented, clinical practice often reveals a wide range of individual responses. Clinicians and researchers have long questioned whether specific patient characteristics—such as age, sex, or baseline metabolic status—influence the degree of weight loss achievable with GLP-1 RA therapy. This systematic review and meta-analysis addresses these uncertainties by quantifying how patient demographics and baseline clinical markers impact weight loss outcomes.
Study Design and Systematic Methodology
Researchers conducted a rigorous systematic review and meta-analysis, querying MEDLINE, Embase, and the Cochrane Central Register of Controlled Trials from their inception through July 26, 2024. The study focused on randomized clinical trials (RCTs) that compared GLP-1 RAs (semaglutide, liraglutide, exenatide, lixisenatide, and dulaglutide) against placebos or other active medications.Of the 7,705 unique records initially identified, 41 articles representing 64 RCTs met the inclusion criteria. The total study population was substantial, with an average trial size of 1,181 participants. The majority of these trials (98.1%) were parallel and multicenter in design. Semaglutide was the most frequently studied agent (43.8% of trials), followed by dulaglutide (18.8%).Data extraction focused on change in body weight, measured either in kilograms or as a percentage change from baseline. The researchers assessed the risk of bias using the Cochrane tool and employed random-effects models for the meta-analysis to account for potential variations across study designs and populations.
Key Findings: The Impact of Sex on Weight Loss
The most striking finding of this meta-analysis was the significant difference in weight loss outcomes between men and women. In an analysis of six trials involving nearly 20,000 patients, women achieved a mean weight loss of 10.9% (95% CI, 7.0%-14.8%). In contrast, men achieved a mean weight loss of 6.8% (95% CI, 4.6%-9.0%).
Statistical and Clinical Significance
This disparity represents a clinically meaningful difference in treatment response. While both sexes benefit significantly from GLP-1 RA therapy, the approximately 4% greater weight loss in women suggests that biological or behavioral factors associated with sex may enhance the pharmacodynamic response to these medications. This finding is particularly relevant for setting patient expectations and tailoring therapeutic goals in clinical practice.
Consistency Across Other Subpopulations
Beyond sex, the study investigated several other potential drivers of treatment heterogeneity. Interestingly, the meta-analysis found no significant differences in weight loss efficacy across the following categories:
Age and Baseline BMI
Analysis of trials involving over 4,300 patients showed that age did not significantly alter the effectiveness of GLP-1 RAs. Similarly, across 15 trials, baseline body mass index (BMI) did not serve as a predictor for the magnitude of weight loss, suggesting that these drugs are equally effective for patients across the spectrum of overweight and obesity classes.
Race and Ethnicity
The study examined data from over 25,000 patients categorized by race and over 8,000 patients categorized by ethnicity. The results indicated that the weight-loss benefits of GLP-1 RAs are consistent across diverse racial and ethnic groups. This is a reassuring finding for public health, indicating that these therapies can be applied broadly without concerns regarding reduced efficacy in specific populations.
Baseline HbA1c
Whether a patient started with high or near-normal hemoglobin A1c levels did not significantly impact the weight loss achieved. This suggests that the weight-modifying mechanisms of GLP-1 RAs operate independently of baseline glucose control levels.
Expert Commentary and Mechanistic Insights
The finding that women lose more weight on GLP-1 RAs than men warrants deeper investigation into the biological plausibility of such a difference. Several theories may explain this heterogeneity:
Biological and Hormonal Factors
Differences in body composition, specifically the ratio of adipose tissue to lean muscle mass, may influence the distribution and metabolism of GLP-1 RAs. Furthermore, interactions between estrogen and the GLP-1 signaling pathway have been suggested in preclinical models, potentially sensitizing the central nervous system’s satiety centers to the effects of the drug.
Behavioral and Adherence Patterns
Some researchers suggest that differences in dietary patterns or adherence to lifestyle modifications during clinical trials may vary by sex. However, because the results were consistent across multiple high-quality RCTs, a biological underpinning remains the most likely primary driver.
Clinical Implications for Personalized Obesity Care
For clinicians, these results underscore the importance of a personalized approach to obesity management. While men still achieve significant weight loss (6.8%), they may require more intensive lifestyle support or higher dosages to reach the same weight-loss milestones as women. Conversely, women may reach their target weight loss more rapidly, which could influence the duration of titration phases or the choice of maintenance doses.
Study Limitations
Despite the robust nature of the meta-analysis, some limitations exist. Only a subset of the 64 trials provided data stratified by sex, race, or ethnicity. Additionally, the analysis focused on GLP-1 RAs as a class; while semaglutide and dulaglutide were well-represented, other agents had less granular data available for subgroup analysis. Future research should prioritize the inclusion of diverse populations and standardized reporting of subgroup outcomes to further refine our understanding of HTE.
Summary and Conclusion
This systematic review and meta-analysis provides critical evidence that GLP-1 receptor agonists are highly effective weight-loss tools with broad applicability. The definitive finding that women experience greater weight loss than men is a pivotal insight for clinical decision-making and patient counseling. Importantly, the lack of heterogeneity across age, race, and baseline BMI reinforces the versatility of this therapeutic class. As we move toward a more personalized model of healthcare, these data offer a foundational step in optimizing obesity treatment for all patients.
Reference
Alexander GC, Xiao X, Dilek S, Lewis S, Deng Q, Kim M, Bolanle D, Saldanha IJ, Mehta HB. Heterogeneity of Treatment Effects of Glucagon-Like Peptide-1 Receptor Agonists for Weight Loss in Adults: A Systematic Review and Meta-Analysis. JAMA Intern Med. 2026 Mar 2. doi: 10.1001/jamainternmed.2025.8222. Epub ahead of print. PMID: 41770554.