Highlights
- Identification of germline and somatic L858R EGFR variants as the cause of a distinct syndrome involving generalized acanthosis nigricans (AN) and pulmonary pathology.
- Clinical hallmarks include early-onset flexural skin thickening, periorificial hyperpigmentation, woolly hair, and palmoplantar keratoderma.
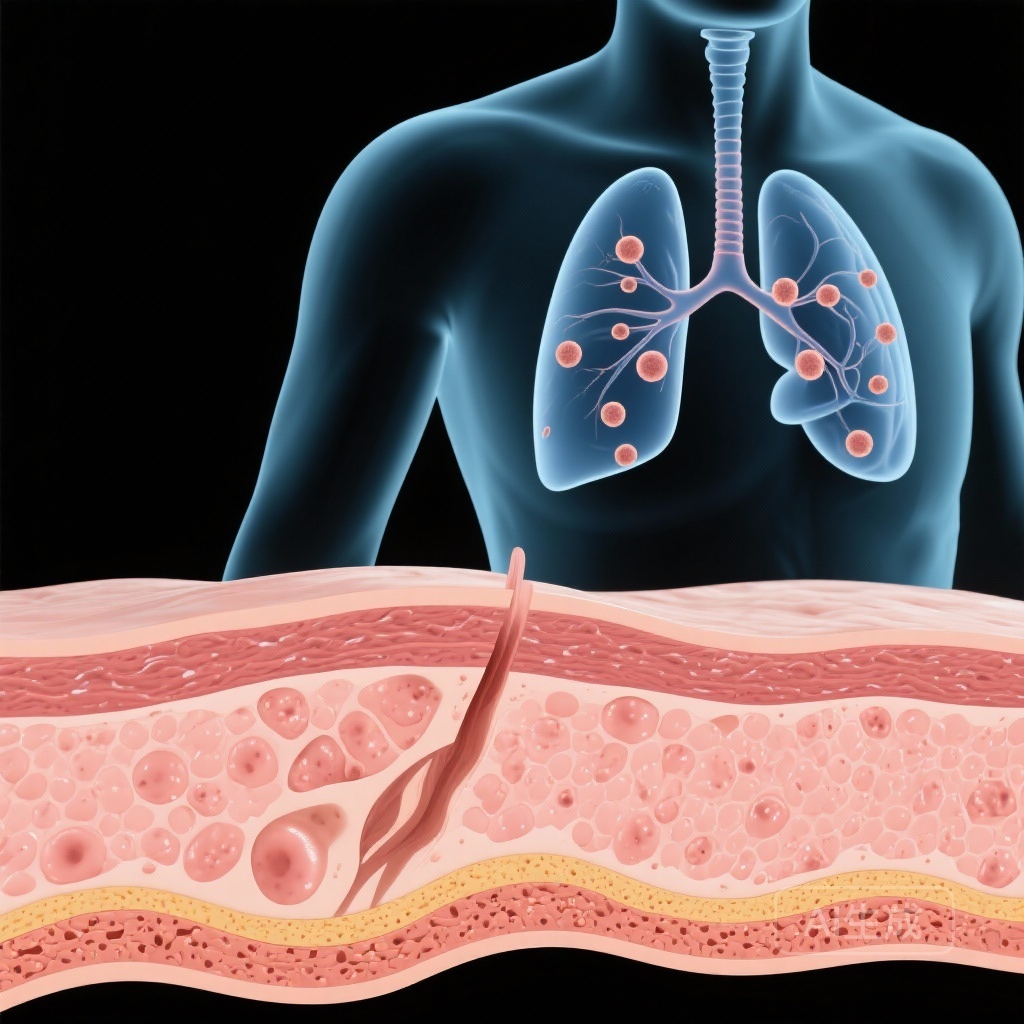
- Detection of pulmonary nodules in affected individuals, which represent potential precursor lesions for lung adenocarcinoma.
- Demonstration of near-complete resolution of both cutaneous and pulmonary symptoms following systemic treatment with EGFR tyrosine kinase inhibitors (TKIs).
Background
Acanthosis nigricans (AN) is a common dermatologic marker typically associated with insulin resistance, obesity, and endocrine disorders. However, when AN presents early in life, is generalized, or occurs in the absence of hyperglycemia, it may signal an underlying genetic syndrome or internal malignancy. While gain-of-function variants in the epidermal growth factor receptor (EGFR) are well-documented drivers of non-small cell lung cancer (NSCLC), their role in causing systemic, syndromic skin disease has remained poorly defined until recently.
The EGFR pathway is critical for keratinocyte proliferation and differentiation. Overactivation of this pathway through somatic mutations is a hallmark of oncogenesis, but de novo germline mutations—specifically the L858R substitution in exon 21—present a unique clinical challenge. This study investigates the genetic basis of severe, early-onset syndromic AN and explores the therapeutic potential of pathogenesis-directed therapy using EGFR inhibitors.
Study Design
This case series study involved three participants with distinct clinical presentations of severe skin thickening and hyperpigmentation. Two individuals presented with normoglycemia and early-onset periorificial hyperpigmentation that progressed to generalized flexural skin thickening. The third individual was initially diagnosed with a widespread epidermal nevus. The age of the participants ranged from 8 to 18 years.
The research team employed a multi-disciplinary approach to characterize the disease:
-
Genetic Analysis:
Whole-exome sequencing (WES) was performed on blood and skin samples to identify pathogenic variants.
-
Molecular Validation:
Studies were conducted on affected skin tissues and cultured keratinocytes to assess EGFR pathway activation, specifically looking at downstream signaling molecules like MAPK.
-
Clinical Assessment:
Participants underwent comprehensive pulmonary function testing and high-resolution lung imaging (CT scans) to screen for systemic involvement.
-
Therapeutic Intervention:
The study monitored the clinical response of these patients to systemic EGFR inhibitor therapy, assessing changes in skin texture and pulmonary nodule burden.
Key Findings
Genomic and Molecular Drivers
All three participants were found to carry the EGFR L858R variant. In the two cases with generalized skin involvement, the variant was de novo germline, while the individual with the localized epidermal nevus presentation exhibited a somatic mosaic variant. Analysis of lesional skin and cultured keratinocytes confirmed a significant increase in EGFR pathway activity. In vitro pharmacologic inhibition successfully suppressed this overactivation, providing a rationale for systemic treatment.
Phenotypic Manifestations
The clinical presentation was remarkably consistent across the germline cases. Key features included:
- Generalized acquired keratoderma with significant accentuation at flexural sites (axillae, neck, groin).
- Woolly hair texture and palmoplantar keratoderma.
- Early-onset periorificial hyperpigmentation.
Crucially, the study identified significant pulmonary involvement. Lung imaging revealed the presence of multiple pulmonary nodules in the participants. Given that the L858R variant is a known driver of lung adenocarcinoma, these nodules are considered precursor lesions, placing these young patients at an exceptionally high lifetime risk for pulmonary malignancy.
Therapeutic Efficacy of EGFR Inhibition
The transition from molecular discovery to clinical application yielded dramatic results. Patients treated with systemic EGFR inhibitors (such as erlotinib or gefitinib) showed significant regression of skin disease. More importantly, follow-up imaging demonstrated a resolution or a substantial reduction in the number and size of pulmonary nodules. This suggests that TKIs can serve as both a therapeutic and a prophylactic measure in this patient population.
Expert Commentary
This research redefines our understanding of the relationship between EGFR signaling and skin physiology. In the context of clinical medicine, the identification of the “EGFR-AN syndrome” provides a critical diagnostic framework for pediatric dermatologists and oncologists. When a clinician encounters generalized AN in a patient with normal glucose metabolism, genetic screening for EGFR variants should be considered mandatory.
The mechanistic link between the L858R variant and the observed phenotype is biologically plausible. EGFR signaling drives the expansion of the stratum spinosum and inhibits terminal differentiation, leading to the hyperkeratotic and acanthotic features of the skin. In the lungs, the same mutation promotes the clonal expansion of alveolar epithelial cells, leading to nodule formation. The success of TKI therapy in this study highlights the power of precision medicine: by targeting the specific molecular driver, clinicians can treat both the visible skin disease and the invisible, life-threatening pulmonary precursors.
Conclusion
The study by Jiang et al. establishes that de novo germline L858R EGFR variants cause a multi-system syndrome characterized by generalized acanthosis nigricans and a high risk of pulmonary disease. This discovery shifts the diagnostic paradigm for severe AN from a metabolic focus to a genomic one. Early recognition is vital, as it allows for the initiation of EGFR-targeted therapy, which has shown remarkable efficacy in reversing skin manifestations and potentially preventing the progression of pulmonary nodules to invasive adenocarcinoma. Further long-term studies are needed to determine the optimal duration of TKI therapy and the long-term prognosis for these individuals.
References
Jiang X, Jeng MY, Yang Z, et al. De Novo Germline L858R EGFR Variants and Generalized Acanthosis Nigricans. JAMA Dermatol. Published online January 14, 2026. doi:10.1001/jamadermatol.2025.5414