Understanding Hypertrophic Cardiomyopathy: A Complex Genetic Puzzle
Hypertrophic cardiomyopathy (HCM) is recognized as the most common inherited cardiac disorder, affecting approximately 1 in 500 individuals worldwide. It is characterized by the unexplained thickening of the heart muscle, specifically the left ventricle and the interventricular septum. This thickening, or hypertrophy, can obstruct blood flow from the heart to the rest of the body, leading to symptoms such as chest pain, shortness of breath, palpitations, and in the most tragic cases, sudden cardiac death in young adults and athletes. For decades, the primary focus of HCM research has been on the sarcomere—the basic contractile unit of muscle cells. Mutations in genes encoding sarcomeric proteins, such as MYH7 and MYBPC3, are well-documented causes of the disease. However, a significant clinical challenge remains: nearly half of all patients diagnosed with HCM do not possess identifiable mutations in these sarcomere genes. This genetic heterogeneity suggests that other, non-genetic or secondary mechanisms must be at play, converging on a common pathway that causes the heart muscle to thicken regardless of the initial trigger. Identifying these common pathways is essential for developing universal therapies that can treat all HCM patients, not just those with specific genetic defects.
The Discovery of CRLF1: A New Player in Heart Health
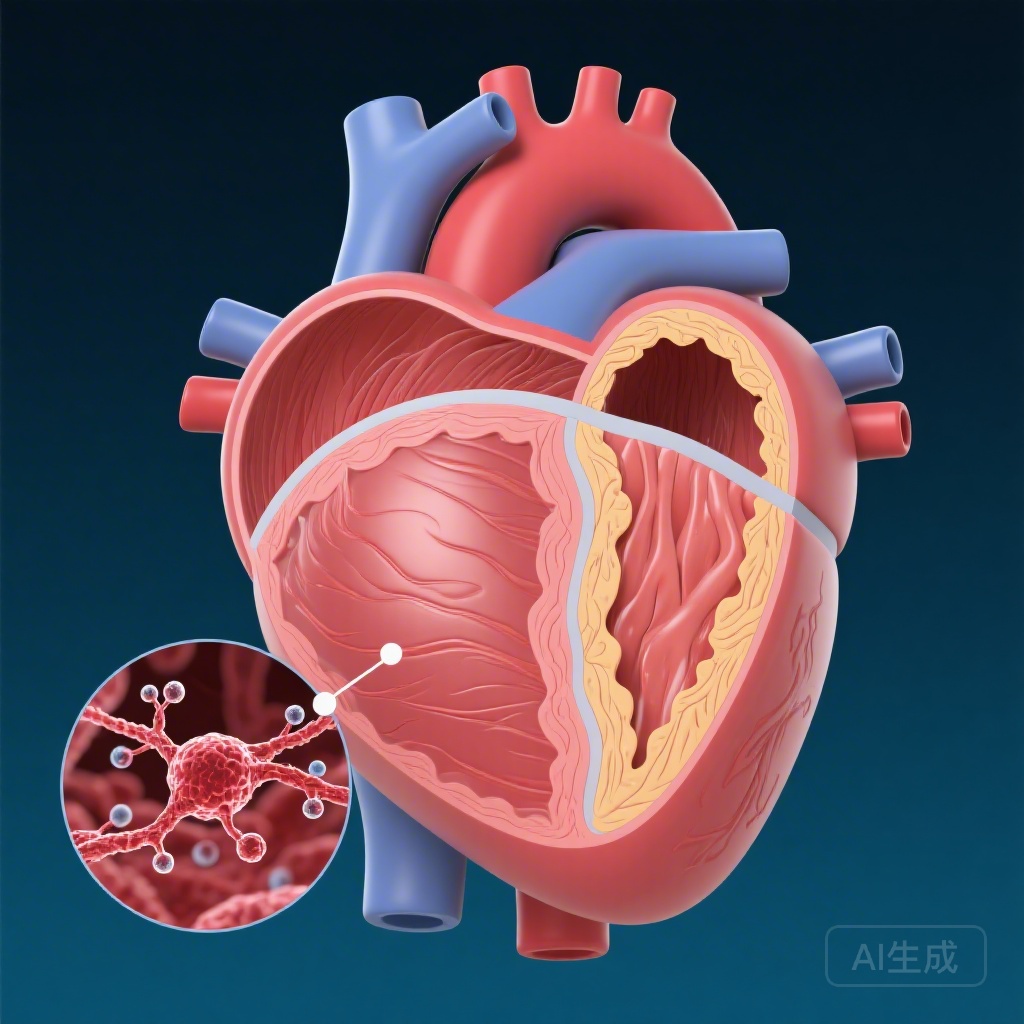
A recent landmark study published in the journal Circulation has shed light on this mystery. Researchers conducted an extensive analysis of hypertrophied heart tissue from 269 patients who underwent surgical myectomy—a procedure to remove excess heart muscle—due to obstructive HCM. By utilizing advanced techniques like bulk RNA sequencing and weighted gene coexpression network analysis (WGCNA), the team sought to identify molecules that were consistently overactive in diseased hearts. The study identified CRLF1 (cytokine receptor-like factor 1) as a major candidate driver of the disease. CRLF1 was found to be significantly elevated not only in the heart tissue of these patients but also in their circulating blood. Most importantly, the research pinpointed the source of this protein: it is predominantly secreted by activated cardiac fibroblasts. Fibroblasts are cells in the heart responsible for maintaining the structural framework of the organ. In HCM, these cells become hyperactive and start secreting CRLF1, which then acts on neighboring cardiomyocytes (heart muscle cells) to trigger growth and thickening.
Mechanisms of Hypertrophy: The CRLF1 and JAK-STAT Pathway
The study describes a paracrine mechanism—a form of cell signaling where one cell produces a signal to induce changes in nearby cells. In this case, cardiac fibroblasts send a CRLF1 signal to cardiomyocytes. Once CRLF1 reaches the muscle cells, it binds to a specific receptor complex known as LIFR (leukemia inhibitory factor receptor). This binding initiates a series of internal signals involving enzymes called Janus kinases (JAK1 and JAK2), which eventually activate STAT3 (signal transducer and activator of transcription 3). When STAT3 is activated, it moves into the cell nucleus and turns on genes that promote cell growth and protein synthesis, directly leading to the enlargement of the heart muscle cells. This pathway represents a common denominator in HCM. Whether the patient has a sarcomere mutation or an unknown genetic cause, the CRLF1-mediated signaling appears to drive the progression of the disease. This discovery is pivotal because it shifts the focus from the muscle cells themselves to the surrounding support cells—the fibroblasts—and the proteins they secrete.
Validation through Cross-Species Research
To confirm these findings, the researchers utilized a mouse model (Myh6 R404Q/+) that mimics human HCM. They observed the same elevation of CRLF1 in the mice as they did in human patients. To test if CRLF1 was truly the cause of the problem, they performed genetic experiments to delete the Crlf1 gene specifically in fibroblasts. The results were remarkable: mice without CRLF1 in their heart fibroblasts showed significantly less heart thickening and improved cardiac function. Furthermore, the researchers tested pharmacological inhibitors designed to block the downstream JAK-STAT pathway. These drugs, which are already used or being studied for other medical conditions, were able to attenuate the HCM symptoms in the animal models. This provided strong evidence that blocking this signaling chain could be a viable strategy for treating human patients.
Clinical Implications and the Future of HCM Treatment
The identification of CRLF1 as a secreted paracrine factor has several major implications for the future of cardiology. First, it offers a potential new diagnostic tool. Since CRLF1 levels are elevated in the circulation of HCM patients, a simple blood test might one day help doctors assess the severity of the disease or monitor its progression. Second, and perhaps most importantly, CRLF1 represents a promising therapeutic target. Current treatments for HCM, such as beta-blockers, calcium channel blockers, and the recently approved mavacamten, primarily focus on managing symptoms or reducing the force of heart contractions. While effective for many, they do not necessarily stop the underlying process of muscle thickening. A therapy that targets CRLF1 or the LIFR-JAK-STAT pathway could potentially halt or even reverse the hypertrophy itself. Because CRLF1 appears to be a universal driver across different genetic backgrounds, such a treatment could benefit the entire spectrum of HCM patients, including the 50 percent who currently lack a clear genetic diagnosis.
Conclusion: A Shift in the Treatment Paradigm
The discovery of CRLF1-mediated signaling marks a significant advancement in our understanding of hypertrophic cardiomyopathy. By revealing that cardiac fibroblasts play a central role in driving muscle growth through secreted factors, this research opens the door to a new era of ‘non-sarcomeric’ therapies. As we move forward, the development of targeted inhibitors against CRLF1 or its receptors could provide a much-needed lifeline for patients living with this complex and often dangerous heart condition. The transition from identifying genetic mutations to understanding the common protein signals between cells represents a vital shift in medical science, bringing us closer to a universal cure for one of the world’s most common inherited heart diseases.