The Hidden Burden of Post-Tuberculosis Lung Disease
While global efforts have successfully increased the rates of tuberculosis (TB) cure, a secondary crisis is emerging: the long-term respiratory morbidity experienced by survivors. Post-tuberculosis lung disease (PTLD) is a heterogeneous condition characterized by chronic respiratory impairment following the completion of anti-tuberculosis therapy. Despite its high prevalence, the underlying biological mechanisms that dictate whether a patient develops a restrictive or obstructive pattern of impairment have remained largely elusive. A landmark prospective study conducted in Nairobi, Kenya, and published in the American Journal of Respiratory and Critical Care Medicine, provides critical insights into the clinical and transcriptomic risk factors that define these distinct PTLD phenotypes.
Study Methodology: A Longitudinal Approach in Nairobi
The researchers conducted a prospective, observational cohort study involving 205 adults with newly diagnosed pulmonary tuberculosis. Participants were enrolled at diagnosis (month 0) and followed through six months of standard anti-tuberculosis therapy. The primary endpoint was the presence of PTLD, defined as abnormal spirometry at 12 months (six months after treatment completion). The study categorized PTLD into two primary phenotypes: restrictive-pattern impairment and obstructive-pattern impairment. To bridge the gap between clinical observation and molecular pathology, the team performed transcriptomic analyses on blood samples collected at diagnosis and at the end of treatment.
Defining the Phenotypes: Clinical Risk Factors
Of the 205 participants, 103 (50.2%) were diagnosed with PTLD at the 12-month mark. This high incidence underscores the fact that microbiological cure does not equate to functional recovery. Among those with PTLD, 60 exhibited a restrictive pattern, while 43 showed an obstructive pattern. Clinical evaluations revealed that participants with PTLD generally had lower mid-upper arm circumference (MUAC) and reduced cough peak flow, suggesting that nutritional status and mechanical lung clearance are early indicators of poor outcomes.
Multivariable analyses identified radiographic severity at the time of diagnosis as a potent predictor for both phenotypes. Each additional lung quadrant involved on a chest radiograph was associated with a more than two-fold increase in the risk of developing both restrictive-PTLD (aOR 2.1, P < .001) and obstructive-PTLD (aOR 2.2, P < .001). Notably, a history of prior tuberculosis was specifically and strongly associated with obstructive-PTLD, carrying an adjusted odds ratio of 5.4 (P < .001), suggesting that cumulative lung insults favor airway remodeling over parenchymal restriction.
Transcriptomic Profiling: Deciphering the Biological Divergence
The most significant contribution of this study is the identification of unique transcriptional signatures that differentiate the two PTLD phenotypes. These molecular markers suggest that restrictive and obstructive diseases are not merely different points on a spectrum, but rather the results of distinct immunopathological processes.
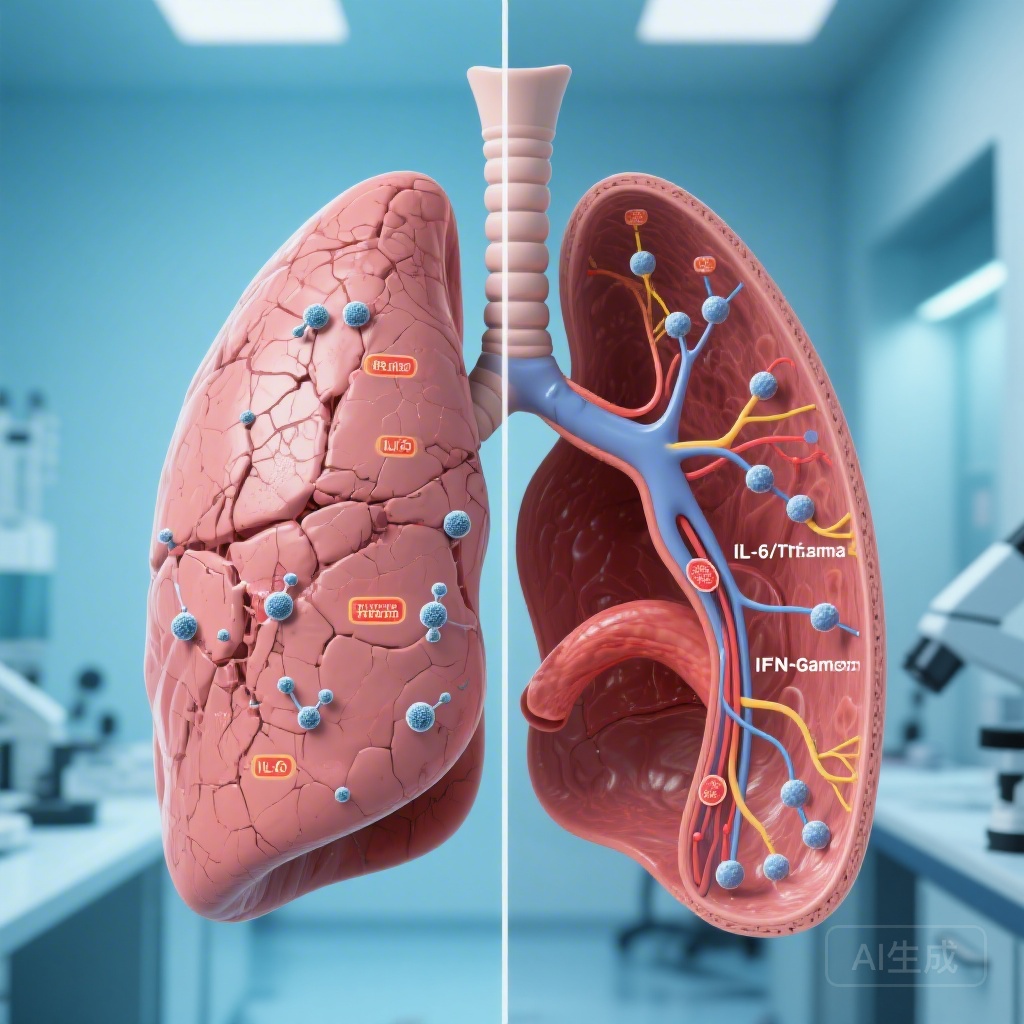
Restrictive PTLD: The Pro-Fibrotic Signature
In participants who developed restrictive-PTLD, transcriptomic analysis at the time of diagnosis showed a significant upregulation of the IL-6/JAK/STAT3 and TNF-alpha signaling pathways. These pathways are classic drivers of pro-inflammatory and pro-fibrotic responses. The presence of these signatures at diagnosis indicates that the trajectory toward restrictive impairment—characterized by reduced lung volumes and parenchymal scarring—is set in motion very early in the course of the infection. This suggests a therapeutic window at the start of TB treatment where anti-fibrotic or targeted anti-inflammatory interventions might mitigate long-term damage.
Obstructive PTLD: The Persistence of Interferon Signaling
Conversely, obstructive-PTLD was characterized by a different temporal and molecular profile. While the restrictive signature was dominant at diagnosis, the obstructive signature became prominent at the end of treatment (month 6). Participants with obstructive-PTLD exhibited transcriptomic upregulation of IFN-alpha and IFN-gamma signaling responses. This suggests that obstructive disease is driven by persistent, unresolved inflammation that continues even after the pathogen has been cleared. This chronic interferon-driven environment likely contributes to airway remodeling and the development of fixed airflow obstruction similar to that seen in chronic obstructive pulmonary disease (COPD).
Expert Commentary: Moving Toward Precision Management
The findings from the Nairobi cohort have profound implications for the management of TB survivors. Historically, PTLD has been treated as a monolithic entity, often managed with generic bronchodilators or pulmonary rehabilitation. However, the divergence in transcriptomic signatures suggests that a stratified approach is necessary. For patients showing early signs of restrictive disease (e.g., high radiographic quadrant involvement and early IL-6 elevation), the introduction of host-directed therapies targeting the JAK/STAT3 pathway could potentially preserve parenchymal integrity.
For those at risk of obstructive disease, particularly those with a history of prior TB, the focus may need to shift toward resolving persistent interferon-mediated inflammation. The study highlights that clinical predictive models are significantly improved when gene expression data is included, pointing toward a future where molecular diagnostics could help clinicians identify high-risk patients before permanent lung damage occurs. One limitation of the study is its reliance on peripheral blood transcriptomics to reflect pulmonary pathology; however, the strong correlation with clinical phenotypes suggests that systemic markers are indeed robust proxies for lung-specific processes.
Conclusion: A New Paradigm for TB Recovery
Post-tuberculosis lung disease is a major public health challenge that affects half of all TB survivors in high-burden settings. By demonstrating that restrictive and obstructive phenotypes are driven by early pro-fibrotic inflammation and persistent interferon signaling, respectively, this research moves us closer to a mechanistic understanding of lung injury. Recognizing these distinct biological trajectories is the first step toward developing targeted interventions that can ensure that a cure for tuberculosis also means a return to full respiratory health.
Funding and Clinical Trial Information
This research was supported by grants from the National Institutes of Health (NIH). Further details on the cohort and data sharing can be found through the American Journal of Respiratory and Critical Care Medicine or via the study’s registration on relevant clinical research databases.
References
1. Zifodya JS, Kijaro LA, Nduba V, et al. Clinical and transcriptomic risk factors for post-tuberculosis lung disease in a cohort of Kenyan adults. Am J Respir Crit Care Med. 2026;41738226.
2. Migliori GB, et al. Post-tuberculosis lung disease: an update on terminology, genetics and management. Eur Respir J. 2021;57(4):2002799.
3. Ravimohan S, et al. Tuberculosis and lung damage: from epidemiology to pathophysiology. Eur Respir Rev. 2018;27(147):170077.